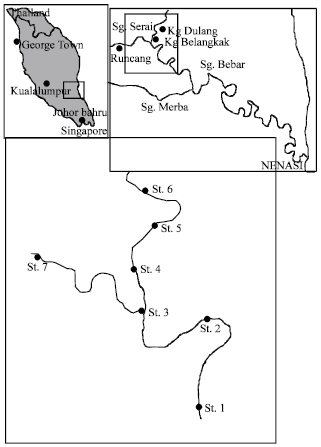
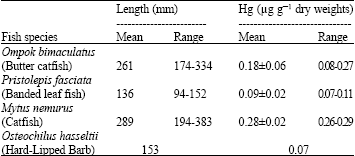
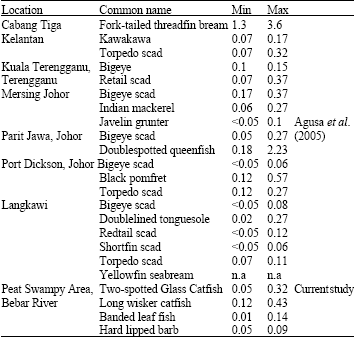
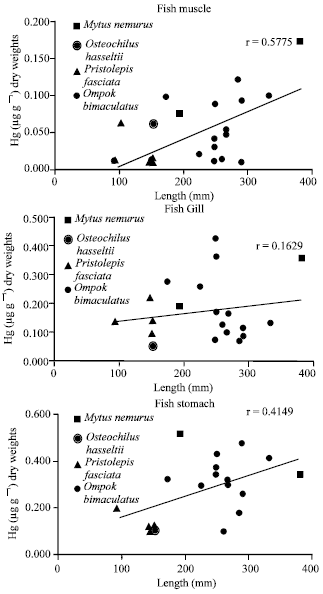
Research Article
Mercury Concentration of Four Dominant Species in the Bebar Peat Swampy Forest River, Malaysia
Institute of Oceanography, Kolej Universiti Sains and Teknologi Malaysia, Kuala Terengganu, Malaysia
M.C. Ong
Institute of Oceanography, Kolej Universiti Sains and Teknologi Malaysia, Kuala Terengganu, Malaysia
A.H. Khali
Peat Swamp Forest Project, UNDP/GEF Funded, Forest Research Institute Malaysia,Kuala Lumpur, Malaysia