Research Article
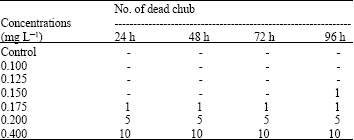
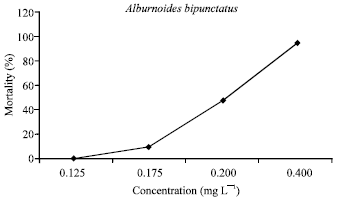
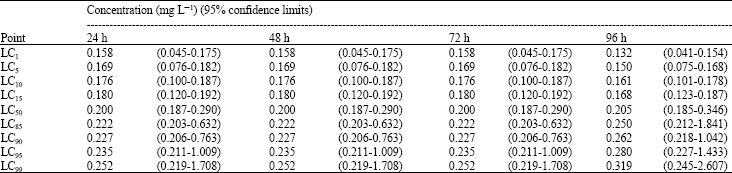
Assessment of Mercuric Chloride Toxicity on Rainbow Trouts (Oncorhynchus mykiss) and Chubs (Alburnoides bipunctatus)
Department of Basic Fisheries Sciences, Faculty of Fisheries and Aquatic Sciences,Rize University, 53100, Rize, Turkey
E. Sibel Besl
Department of Basic Fisheries Sciences, Faculty of Fisheries and Aquatic Sciences,Rize University, 53100, Rize, Turkey
Ilhan Altinok
Department of Fisheries Technology Engineering, Faculty of Marine Sciences,Karadeniz Technical University, 61530, Trabzon, Turkey
Cengiz Mutlu
Department of Biology, Faculty of Art and Sciences, Giresun University, Giresun, Turkey