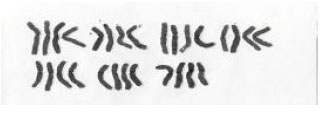Research Article
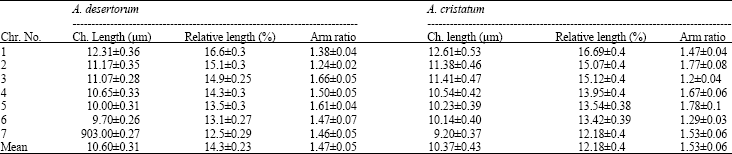
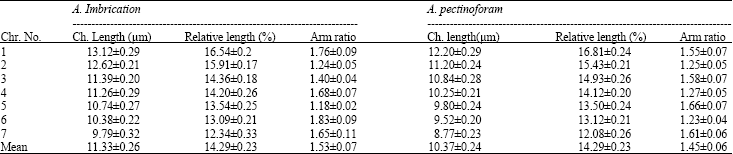
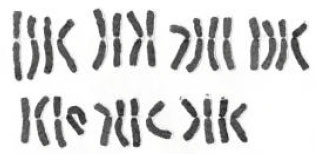
Karyological Study of Four Species of Wheat Grass (Agropyron sp.)
Department of Crop Production and Breeding,Faculty of Agriculture, University of Mohaghegh Ardebili, Ardebil, Iran
Y. Agayev
Agricultural Biotechnology research Institute of Iran, Karaj, Iran
S.A.A. Fathi
Department. of Crop Protection, Faculty of Agriculture,University of Mohaghegh Ardebili, Ardebil, Iran