Research Article
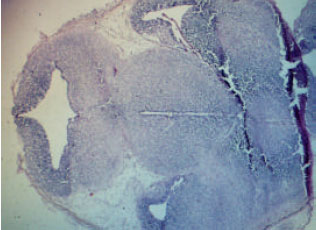
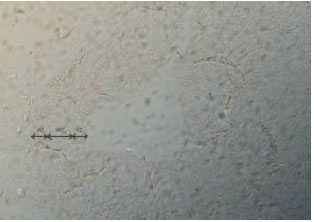
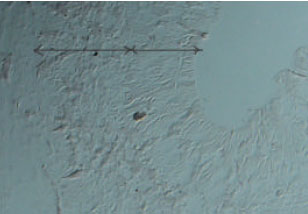
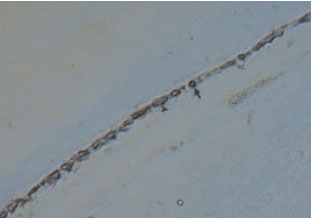
Expression of Neuronal Nitric Oxide Synthase During Embryonic Development of the Rat Optic Vesicle
Department of Anatomy and Histology, Iran University, P.O. Box 14155-6183, Tehran, Iran
S. Majidzadeh
Department of Anatomy and Histology, Iran University, P.O. Box 14155-6183, Tehran, Iran
M. Fattahi
Department of Anatomy and Histology, Iran University, P.O. Box 14155-6183, Tehran, Iran
M. Samadi
Department of Anatomy and Histology, Iran University, P.O. Box 14155-6183, Tehran, Iran
P. Tabatabaeei
Department of Anatomy and Histology, Iran University, P.O. Box 14155-6183, Tehran, Iran