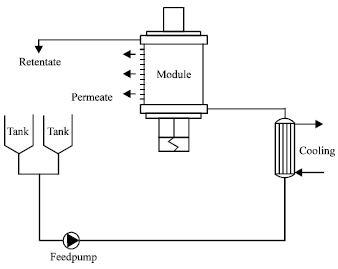
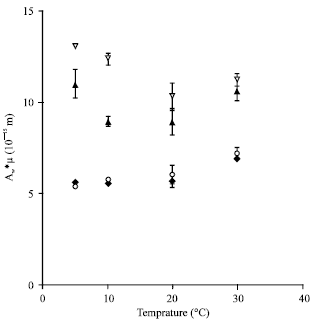
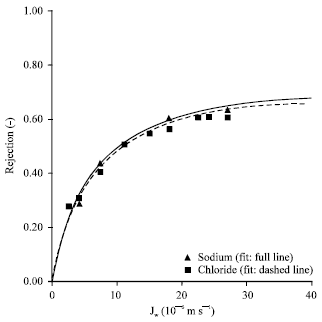
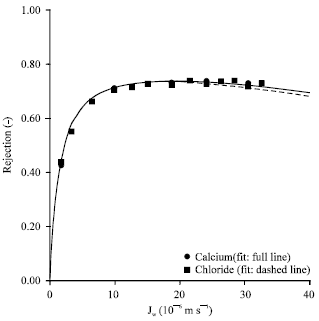
Research Article
Use of Nanofiltration for Concentration and Demineralization in the Dairy Industry
Faculty of Environment, University of Tehran, P.O. Box 14155-6135, Tehran, Iran
Touraj Nasrabadi
Faculty of Environment, University of Tehran, P.O. Box 14155-6135, Tehran, Iran