Research Article
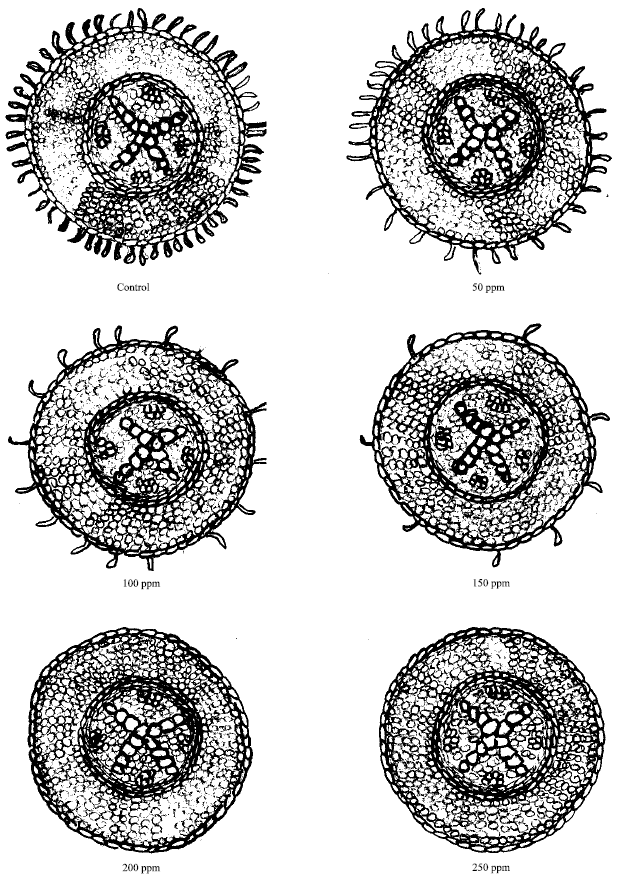
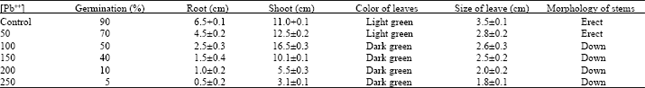
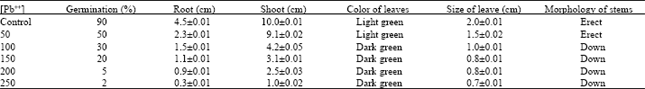
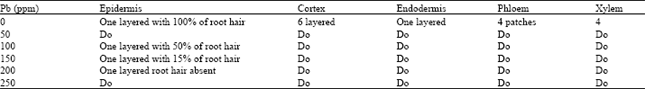
Phytotoxicity of Pb: I Effect of Pb on Germination, Growth, Morphology and Histomorphology of Phaseolus mungo and Lens culinaris
Department of Chemistry, Jinnah University for Women, 5-C Nazimabad, 74600 Karachi, Pakistan
Saba Haider
Department of Botany, Jinnah University for Women, 5-C Nazimabad, 74600 Karachi, Pakistan
Shabana Askari
Department of Botany, Jinnah University for Women, 5-C Nazimabad, 74600 Karachi, Pakistan