Research Article
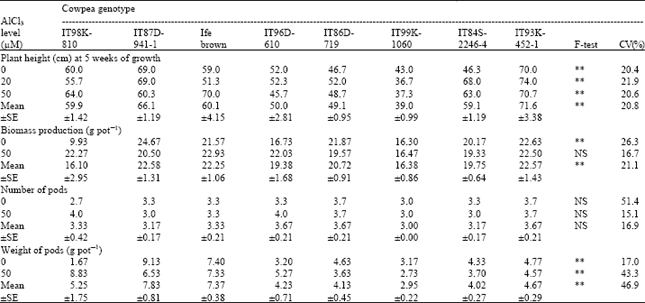
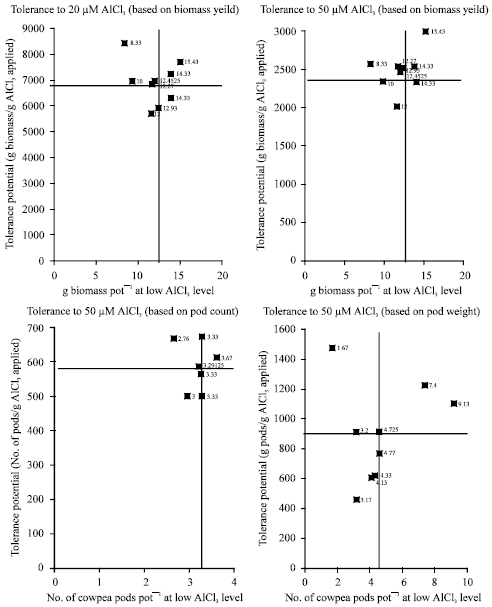
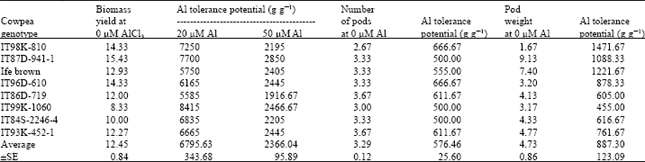
Evaluation of Differences in Tolerance to Aluminium Toxicity among Some Tropical Cowpea (Vigna unguiculata) Genotypes
Department of Agronomy, University of Ibadan, Ibadan, Nigeria
Gunter Neumann
Institut fur Pflanzenernahrung (330), Universitaat Hohenheim, 70593 Stuttgart, Germany