Research Article
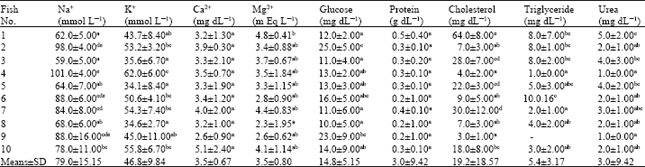
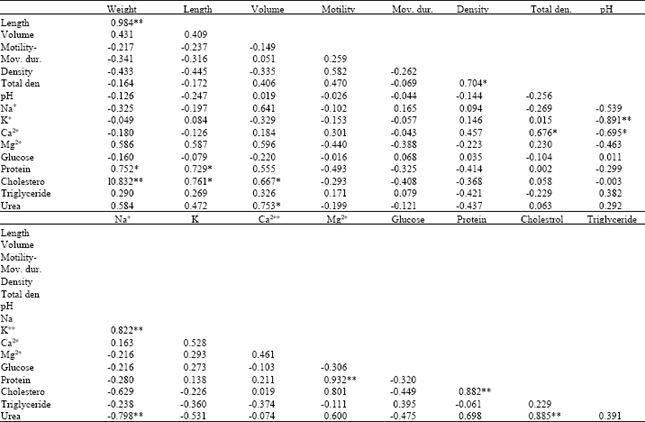
Relationship Between Body Conditon, Physiological and Biochemical Paramaters in Brown Trout (Salmo trutta fario) Sperm
Department of Aquaculture, Faculty of Fisheries, Mustafa Kemal University,31040 Hatay, Turkey
Selcuk Secer
Department of Fisheries and Aquaculture, Faculty of Agriculture, Ankara University, 06110 Ankara, Turkey
Neslihan Bukan
Department of Biochemistry, Faculty of Medicine, Gazi University, 06510 Ankara, Turkey
Ergun Akcay
Deartment of Reproduction and Artificial Insemination, Faculty of Veterinary, Ankara University, 06110 Ankara, Turkey
Necmettin Tekin
Deartment of Reproduction and Artificial Insemination, Faculty of Veterinary, Ankara University, 06110 Ankara, Turkey