Research Article
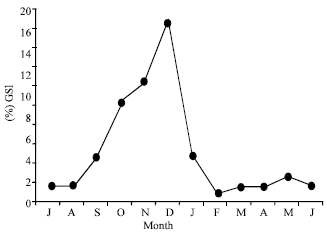
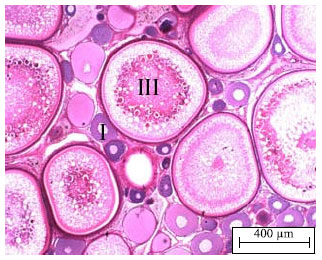
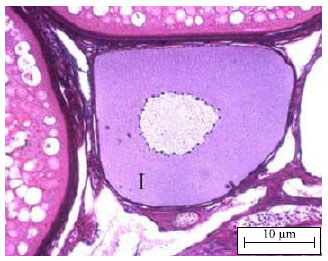
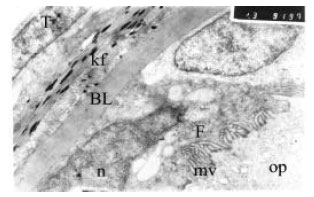
Changing of Follicular Epithelium During Oogenesis in Rainbow Trout (Oncorhynchus mykiss, W.), Studied by Light and Electron Microscopy
Department of Biology, Faculty of Science and Art, Marmara University, G�ztepe, İstanbul, Turley
Y. Aytekin
Department of Histology and Embryology, Faculty of Istanbul Medicine, Istanbul University, �apa, Istanbul, Turkey
R. Yuce
Department of Biology, Faculty of Science and Art, Marmara University, G�ztepe, İstanbul, Turley