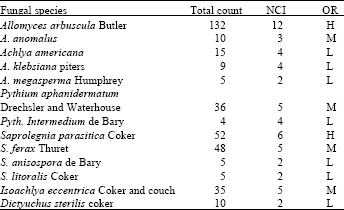
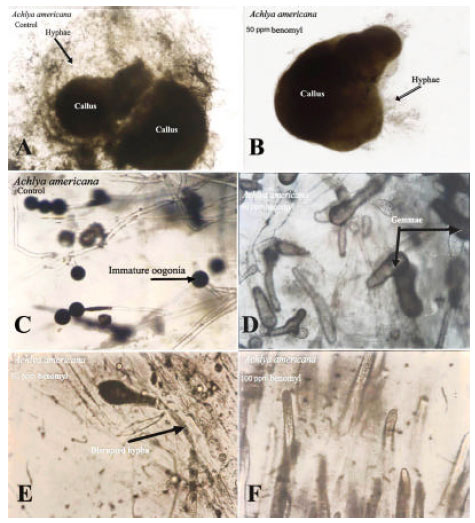
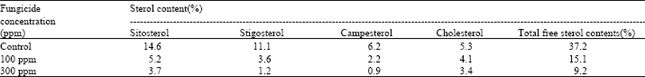
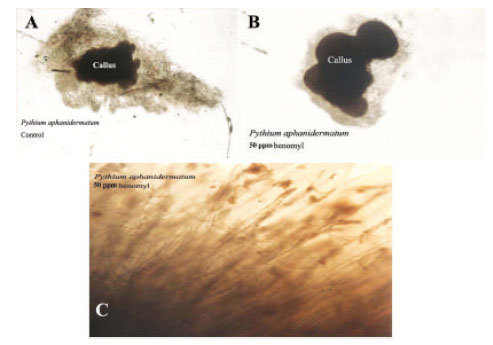
Research Article
Effect of Benomyl Treated Garlic on Growth and Sporulation of Pythium aphanidermatum and Achlya americana
Department of Botany, Faculty of Science, South Valley University, Sohag, Egypt
A.A. Galal
Department of Botany, Faculty of Science, South Valley University, Sohag, Egypt