Research Article
Isolation of Endophytic Fungi from Brucea javanica L. (Merr.) and Cytotoxic Evaluation of their n-butanol Extract from Fermentation Broth
Department of Biomedical Sciences, University of Indonesia, Jl Salemba, Jakarta, Indonesia
Robert Utji
Department of Microbiology, Faculty of Medical, University of Indonesia, Jl Salemba, Jakrata, Indonesia
Pratiwi Sudarmono
Department of Microbiology, Faculty of Medical, University of Indonesia, Jl Salemba, Jakrata, Indonesia
Leonardus B.S. Kardono
Research Center for Chemistry, Indonesian Institute of Sciences, Kawasan PUSPITEK, Serpong, Indonesia









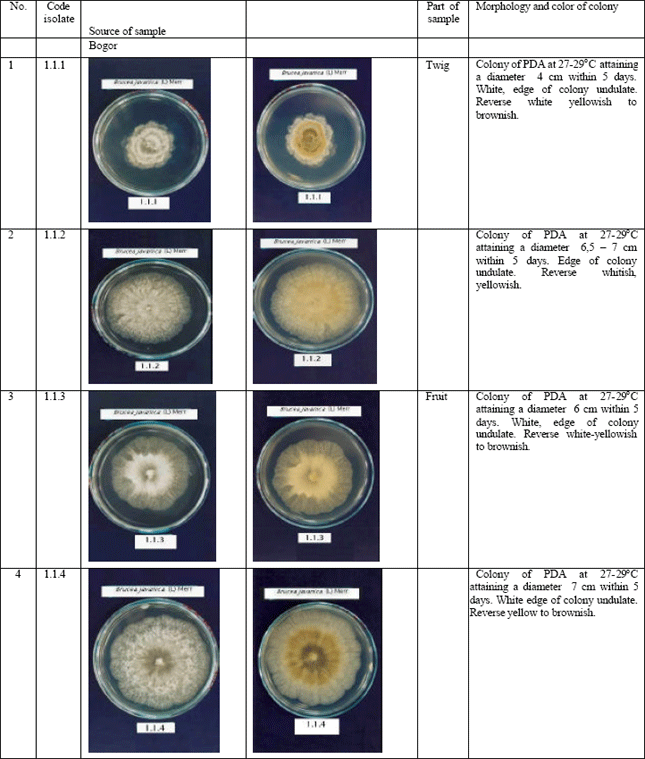
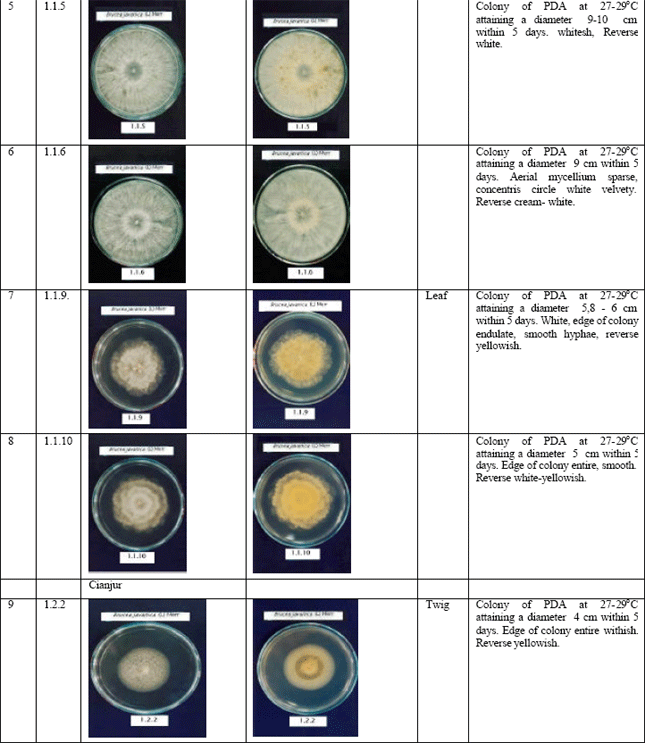
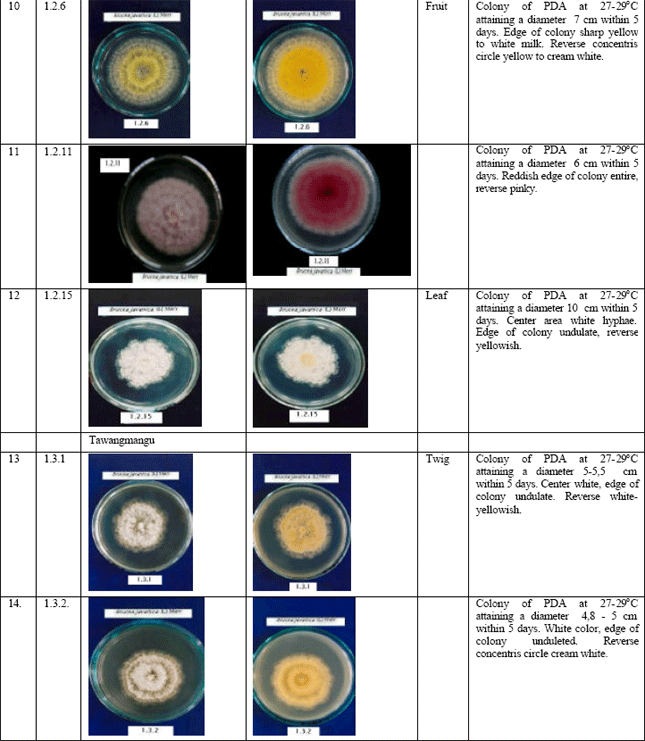
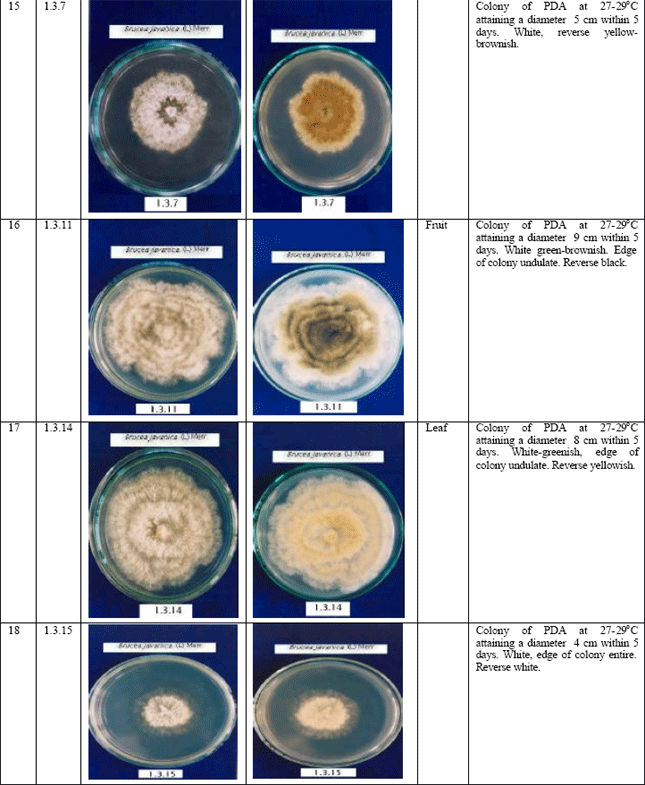
Jennifer Reply
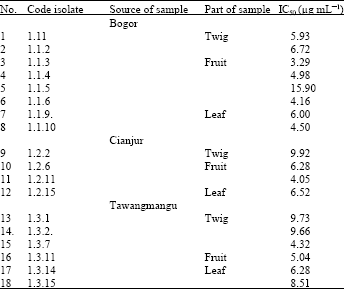
Hello, may i know what is the genus of fungi which is labelled as 1.3.11 (white green brownish,edge of colony undulated, reverse black,part of samples:fruit)?
Look forward to your reply. Thank you.