Research Article
The Proinflammatory Cytokines in Children with Autism
National Research Centre, Dokki, Cario, Egypt
Maysa Tawheed Saleh
National Research Centre, Dokki, Cario, Egypt
The human brain is the second organ that contains the highest concentration of lipids next to the adipose tissue. Phospholipid (PL) fatty acids are major structural components of neuronal cell membranes, which modulate membrane fluidity and hence function. Evidence from clinical and biochemical sources have indicated changes in the metabolism of fatty acids in several psychiatric disorders (Vancassel et al., 2001). In the Central Nervous System (CNS), PL is exceptionally rich in highly unsaturated Fatty Acids (FA). Thus, about 20% of the dry weight of the brain consists of polyunsaturated fatty acids (PUFA) containing at least 20 atoms of carbon, such as arachidonic acid (20: 4n-6, AA) and docosahexaenoic acid (22: 6n-3, DHA) (Vancassel et al., 2001).
A growing body of studies has demonstrated the involvement of PUFA in the regulation of many biochemical events such as neurotransmitter release and uptake, receptor function in the CNS (Murphy, 1990) and enzymatic processes (Bourre et al., 1989).
Autism is a highly prevalent syndrome with a complex etiology. Extensive research has led to the hypothesis that autism is a relating deficiency due to a developmental disorder of the central nervous system was also observed (Trottier et al., 1999). Modifications of lipid metabolism have been described in several psychiatric illnesses (Bennett and Horrobin, 2000), in particular in schizophrenia (Fenton et al., 2000) and in Attention Deficit Hyperactive Disorder (ADHD), (Burgess et al., 2000). These syndromes and autism shared some common behavioral traits, in particular impairment of communication abilities (Peet et al., 1999).
Recent progress in basic immunology has revealed an intricate immune defense network mounted by innate and adaptive immunity. The proinflammatory cytokines produced by innate immunity are known to cross the intact blood-brain barrier and affect hypothalamo-pituitary-adrenocortical (HPA) axis inducing stress responses (Barkhudaran and Dunn, 1999; Wang and Dunn, 1999).
Both ω6 (ω6) and ω3 (ω3) polyunsaturated fatty acids (PUFAs) are potent modulators of the Inflammatory Response System (IRS) and of lymphocytic and monocytic functions (Maes and Smith, 1998). The ω6 PUFAs have proinflammatory capacities: ω6 PUFAs-in particular, C20:4ω6 (arachidonic acid)-are precursors of proinflammatory eicosanoids of the prostaglandin-2 series, such as PGE2 (Smith, 1991) and increase the production of IL-1, TNF-α and IL-6 (Hayashi et al., 1998; Tashiro et al., 1998). The ω3 PUFAs, on the other hand, have anti-inflammatory and immunosuppressive effects (Meydani et al., 1991).
There is now some evidence that autism may be accompanied by abnormalities in the Inflammatory Response System (IRS). Products of the IRS, such as proinflammatory cytokines, may induce some of the behavioral symptoms of autism, such as social withdrawal, resistance to novelty and sleep disturbances (Croonenberghs et al., 2002).
Aim of the present study was to assess the value of the proinflammatory cytokines (interleukins IL-1b and IL-6) and their relation to PUFA in autistic children.
The present study was conducted on 21 autistic children who were fulfilling the criteria for diagnosis of autistic disorders according to the DSM IV diagnostic criteria for research (American Psychiatric Association, 1994). Their age ranged from 4-12 years, 16 were males and 5 were females.
Any history of neurological disorders (i.e., cerebral palsy) or significant medical diseases (i.e., hepatitis or nephritis) were excluded.
For the sake of comparison 21 age and sex matched comparable healthy children who had no history suggesting medical, neurological or psychiatric disorders were studied as control group. Their ages ranged from 4-12 years, they were 13 males and 8 females.
The National Research Center Ethical Committee approved the study. The parents of all subjects gave informed written consent to the participation of their children for measurement of plasma biological parameters. All children were clinically interviewed to fit the diagnostic criteria of the Diagnostic and Statistical Manual-IV.
Full history laying stress on the socio-demographic history, as well as the perinatal history and vaccination were taken. Developmental mile stones, presence of sleep disturbances, convulsions and gastrointestinal problems were included.
Regression was defined as loss of previously acquired language and social interaction skills and was based on family reporting; it was not a selection criterion. Family history of psychiatric disorders was evaluated.
Full clinical examination was done, including neurological examination to detect motor system affection in addition to nutritional deficiency.
Peripheral blood samples were obtained when the study subjects were a febrile, not on antibiotics or steroids and did not have symptoms indicating viral syndrome or other common childhood infection.
Laboratory investigations included
| • | Plasma fatty acid was measured using gas chromatography (Willem et al., 1995). |
| • | Interleukins 1-β (IL-1β) and interleukin-6 (IL-6) were assayed using Enzyme Amplified Sensitivity Immunoassay. |
Sample collection: Five millimeter of venous blood were collected and equally subdivided into two clean dry tubes. The first one contains fluoride EDTA as an anticoagulant, centrifugation of the samples was done immediately, plasma was separated and stored in sterile aliquots at 20°C until assay of PUFAs. The second tube was empty and blood was left to clot, then serum was separated and stored at 20°C until assay of IL-6 and IL-1B.
Determination of plasma PUFAs: The principle was the injection of separated fatty acids from plasma into the gas chromatography instrument. Separation of fatty acids from plasma was done by addition of 3 mL of 3 g L–1 solution of nonanoic acid in 100 μL of acetyl chloride slowly with magnetic stirring for 45 min at room temperature. Three milliter of 6% potassium carbonate solution was added in water with stirring while cooling the mixture in an ice bath. After that, 300 μL of hexan and vortex were added and the sample was cooled at 4°C for 30 min. Then centrifugation was done for 10 min. Finally 100 μL of the upper layer was removed and 1.1 μL was subjected to Gas Chromatography H5890 (Hewlett-Pakard, Palo/Alto,CA), (Sacks, 1999).
Determination of IL-1B and IL-6: IL-1B was done by using Biosource EASIA (a solid phase Enzyme Amplified Sensitivity Immunoassay performed on microtiter plate) an Immunoenzymometric assay for quantitative measurement of human interleukin-1B in serum and other biological fluids. The intra-assay is 2.2% CV and the inter-assay is 4.6% CV (Dinrello, 1988).
IL-6 was done by using Biosource EASIA (a solid phase Enzyme Amplified Sensitivity Immunoassay performed on microtiter plate) an Immunoenzymometric assay for quantitative measurement of human IL-6 in serum and other biological fluids. The intra-assay is 4.6% CV and the inter-assay is 2.2% CV (Moine, 1994).
Statistical analysis: Results were processed and statistics were carried out using statistical package for social sciences (SPSS, version 10). All data were expressed as means±SD. Group means were compared by student t-test, p-value<0.05 was considered significant. Pearson Correlation Coefficient was calculated to measure the strength of association between two quantitative groups.
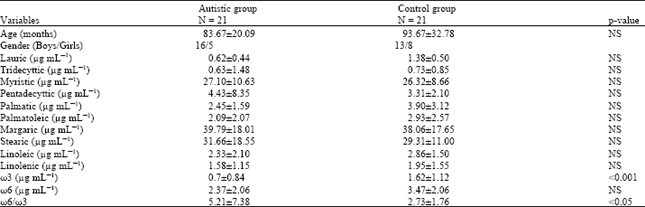
Table 1 shows that the mean age of autistic children was 83.67±20.09 months, while the mean age of the control group was 93.67±32.78 months. The autistic patients were 16 males (76.19%) and 5 females (23.81%), while the control group were 13 males (61.90%) and 8 females (38.10%) (Table 1).
| Table 1: | Comparative studies of serum levels of different fatty acid in different studied groups |
 | |
| NS = Non Significant, p<0.001 = Significant | |
| Table 2: | Plasma polyunsaturated fatty acid in relation to gender |
 | |
| NS = Non Significant (p>0.05), S = Significant (p<0.001) | |
| Table 3: | Comparative study between Interleukin-1B and Interleukin-6 in the studied groups (control, boy, girls) |
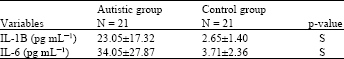 | |
| S = Significant (p<0.001) | |
Plasma PUFAs in different study groups: There was no significant change in the plasma levels of plasma PUFAs except for ω3 when autistic patients compared to the controls (p>0.05). The levels of plasma linoleic acid (18: 2n-6), which is the precursor of the (n-6) series, were identical for both groups of subjects. However, no statistical difference in the total (n-6) PUFA levels was observed between the autistic and the control groups. The major variations observed in PUFA levels between the two groups of subjects involved the (n-3) PUFA series. The results showed that linolenic acid levels (18: 3n-3) were identical in both groups of subjects. However, there was a significant reduction level (n-3) PUFA in autistic children.
The total ratio of ω6/ω3 was statistically significant between the autistic group and the control group (Table 1).
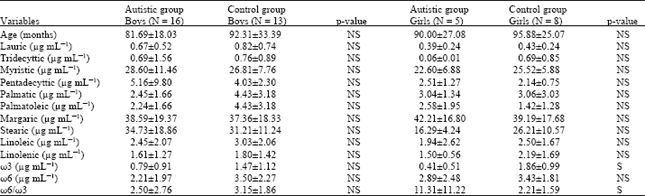
Plasma PUFAS level and gender: In comparison to samples from boys with autism and controls, PUFAs did not show any statistical differences between the two groups, though there was a tendency to lower levels in male cases in linoleic, linolenic as well as ω6 and ω3 (Table 2).
PUFAs did not show any change in girls with autism when compared to controls, except for a significant reduction in ω3 levels (p<0.001), as well as in ω6/ω3 (p<0.005), (Table 2).
Comparison did not show any significant differences in plasma PUFAs, ω3, ω6 PUFAs, or the ω6/ω3 PUFA ratio between male and female.
Serum interleukin-1B and interleukin-6 in different studied groups: Significant higher levels of both IL-1B and IL-6 were observed in autistic patients as compared to the control (Table 3).
A significant increased level was observed between autistic boys and autistic girls when compared to their peers of the control group. Meanwhile, no statistical change was noted between boys and girls of the autistic group (Table 3).
Interleukins in relation to PUFAs: A significant increase in IL-1b and IL-6 in autistic group coincides with a significant reduction in the level of ω3 and slight reduction in ω6 as well as significant higher level of ω6/ω3 PUFA ratio.
Serum IL-1b and IL-6 did not correlate with any of the studied PUFAs. However there was an association between the level of IL-6 and ω3 in female cases.
Little is known about the etiology of autism. Abnormalities in the fatty acid compositions of phospholipids, the major constituents of cell membranes, have been implicated in several neurodevelopmental disorders that manifest with psychiatric symptoms. For example, in schizophrenia, changes of Red Blood Cell (RBC) membrane phospholipids such as deficiencies in n-3 polyunsaturated fatty acids (PUFA) have been reported (Peet et al., 1996; Richardson et al., 2000).
Similarly, defects of fatty acids and phospholipids have recently been reported in autism subjects, including not only reduced levels of n-3 PUFA, but also increased levels of Saturated Fatty Acids (SFA) in the RBC membrane (Bell et al., 2000, 2004). Further evidence from Bell et al. (2004) suggested that decreased levels of arachidonic acid (ARA), docosatetraenoic acid (DTA) and docosahexaenoic acids (DHA) in RBC membranes from autism subjects could be caused by increased activity of RBC type IV phospholipase A2, suggesting that altered metabolism of phospholipids may occur in autism (Bell et al., 2004). In addition, some of the problems that have been reported in autism, such as excessive oxidation or decreased dietary supplies of essential fatty acids may contribute to fatty acid deficiencies (Ming et al., 2005).
This study was undertaken to evaluate fatty acid metabolism, which might have a biological and etiological implications in autistic patients. Moreover, we assessed the immune responses in autistic children secondary to frequent parental reports of adverse reactions to benign environmental factors including childhood infection. Furthermore, we evaluated the relation between these levels and the plasma level of fatty acids in these patients.
In view of the vital role of fatty acids in neural membrane function and cell homeostasis, the defects in fatty acid metabolism may have important biological and etiological implications in autism. We examined the fatty acids in the plasma of a population of autistic subjects compared to controls. This study showed major variations in PUFA levels between the two groups of subjects involved the (ω3) PUFA series. The results showed that linolenic acid levels (18: 3n-3) were identical in both groups of subjects. However, there was a significant reduction level (n-3) PUFA in autistic children. On the other hand, the levels of plasma linoleic acid (18: 2n-6), which is the precursor of the (n-6) series, were identical for both groups of subjects. However, no statistical difference in the total (ω6) PUFA levels was observed between the autistic and the control groups.
Inflammatory cytokines and their receptors are distributed in various brain regions and influence neural development as well as synaptic transmission and behavioral traits (Vereker et al., 2000; Nawa et al., 2000; Yamada et al., 2000). Cytokine induction and/or signaling, which is triggered by abnormal immune reaction or inflammation, can perturb normal brain development, leading to later perceptual and cognitive alterations Gilmore and Jarskog, 1997; Nawa et al., 2000). The higher risk of schizophrenia and autism is associated with the interleukin (IL)-1 genome complex and/or tumor necrosis factor α gene (Katila et al., 1999; Boin et al., 2001).
Inconsistent with this hypothesis, the levels of proinflammatory cytokines (IL-6 and IL-1β) were significantly higher in autistic children than in controls. These proinflammatory cytokines affect hypothalamo-pituitary-adrenocortical (HPA) axis and can potentially influence the functions of the central nervous system (CNS), (Barkhudaran and Dunn, 1999; Wang and Dunn, 1999). Elevations of these cytokines suggest that, the children with autism have a heightened immune response.
Recent epidemiological studies indicate an increase in the prevalence of autism over the past 15-20 years (Fombonne, 1999), which may indicate the role of certain environmental factors. Many parents report the onset of regression autism following immunization and/or benign childhood infection. They also often report aggravation of symptoms following benign viral infection/immunization.
The brain has for many years been considered an immunologically privileged site, suggesting reduced or altered immunological responsivity. Evidence in this regard includes the brain’s lack of adequate lymphatic systems to capture antigens, protection from circulating blood by the blood-brain barrier and failure to exhibit a classic inflammatory response characterized by early invasion of macrophages and leukocytes. Recent developments in neuroimmunology however have challenged some of these concepts (Cserr and Knopf, 1992). The brain can certainly exhibit many of the hallmarks of inflammation in response to infection or injury. They include edema, activation of resident phagocytes (microglia), local invasion of circulating immune cells and production of cytokines. The role of these various cytokines in different brain activities is a topic of intense investigation and debate at the present time.
Interleukin (IL)-1β, the most potent proinflammatory cytokine, has been found to induce stress and anxiety-like behavior in rodents (Song, 2002). This cytokine stimulates the hypothalamus to release Corticotropin-releasing Factor (CRF), which, via adrenocorticotropic hormone, induces the secretion of glucocorticoids from the adrenals. IL-1β also activates central neurotransmitters, thereby increasing the turnover of noradrenaline, serotonin and dopamine. The observed changes are similar to those seen when rodents respond to a stressor (Song et al., 1999).
Abnormalities related to the innate immune response have been reported to occur in children with ASD. Elevated levels of Interleukin 1 receptor antagonist (IL-1RA) have been found, indicative of an increased response by cells of the monocyte-macrophage lineage (Croonenberghs et al., 2002). Elevated levels of the proinflammatory cytokines Tumor Necrosis Factor (TNF)-α and IL-1β were also produced by the PBMC from children with ASD, in both media alone without stimulation and in response to lipopolysaccharide (LPS), a stimulant for cells of monocyte-macrophage lineage (Jyonouchi et al., 2002; Jyonouchi et al., 2001).
The protective effect of PUFAs is mediated by multiple mechanisms, including their antiinflammatory properties. The association of physiological PUFA levels with pro- and antiinflammatory markers has not been established.
Ferrucci et al. (2006) observed that total n-3 fatty acids were associated with lower IL-6 and lower arachidonic and docosahexaenoic acids were associated with significantly higher IL-6. They supported the notion that n-3 fatty acids may be beneficial in patients affected by diseases characterized by active inflammation. In the present study, a significant increase in IL-1b and IL-6 in autistic group coincides with a significant reduction in the level of ω3 and slight reduction in ω6 as well as significant higher level of ω6/ω3 PUFA ratio.
Serum IL-1b and IL-6 did not correlate with any of the studied PUFAs. However there was an association between the level of IL-6 and ω3 in female cases.
We report the results of a case-control study of measured cytokine levels from children with autism and controls. Compared to controls, the cases had evidence of an activated adaptive immune response, predominantly IL-1b and IL-6. These results open up very interesting perspectives for further investigation into this immune regulatory mechanism in autism.