Research Article
Study on Qualitative Change of Spermatozoid on Lori Ram in vitro
Department of Animal Husbandry, Lorestan University, Khorram Abad, Iran
U. K. Doronin
Moscow State University, Russia
S. Kadivar
Medical University of Gilan, Iran
Recognition of morphology, normal and abnormal forms of spermatozoa and their proportion is used to determine fertility criteria. For evaluation of fertility, variables including number of spermatozoa, motility percentage, moving type and different percentages (A, B, C and D), normal spermatozoa percentage, is measured. Considering normal fertility in which percentage of normal spermatozoa should not be less than 85% (Shafei, 2005). Abnormal shapes can have primary or secondary origin. Double twin, double tail, having protoplasmic droplet are related to primary abnormality where as, bent mid piece, shoe hook tail seem related to secondary abnormality. Primary abnormalities are related to organic defect (epydidymis and other genital duct in male reproductive system) In contrast, Secondary abnormalities are originated outside of body. Protoplasmic droplet is a sign of insufficient maturity which originated from epydidymis. Generally genetic factors and nutrition contribute to abnormal shapes (Sankai et al., 2001). Coiled spermatozoa have secondary origin which was named Dag effect by Blom (1966). In samples of rabbit spermatozoa, this shape was reported 9.1% (Kuzmeninsky et al., 1996). So seem in time storing (liquid condition at 4°C) normal shape of spermatozoid be changed. for example detached head, shoe-hook tail, loose cap from head and coiled in mouse spermatozoa was appear (Mohammadzadeh and Doronin, 2005), This being the case in Lorestan province of Iran for artificial insemination liquid semen is used but in process of storing we haven’t know these abnormalities is or not. Do change form of normal spermatozoid? Do these changes similar to other researches? And finally do these quality parameters significantly change? This study was conducted to evaluate abnormal spermatozoa percentage and study on different shapes and their procedure of change in Lori ram spermatozoa.
In this study conducted in Lorestan province of Iran on June 2005. After a period of fattening 10 Lori rams which was under control of Jahad-Keshavarzi of animal husbandry, was selected. After killing, testis's, were cut from spermatical cord and samples was taken according following steps. Deferent duct (final segment of epydidymis to ampulla) cut, vessels and fat separated and its content was extracted and placed in epindorf (1 mL), after adding sodium citrate (3%). Hemocytometer was used to make main solution (1 million mL-1). Sperm motility was determined immediately. Samples was kept at 4°C and was counted. To achieve better evaluation of sperm shapes, samples were stained by Eosin- Nigrozin solution. At first, 5 mL of diluted semen was placed on glass slide then 5 mL Eosin-Nigrozin was added. By using another glass slide was mixed, spread on whole glass slide and dried in oven at 4°C. Finally abnormal shapes were counted and proportion of them determined. Due to slight changes shape of spermatozoa sample analysis was done every month. After evaluating, they analyzed by t-test and graphs was drawn by Excel software.
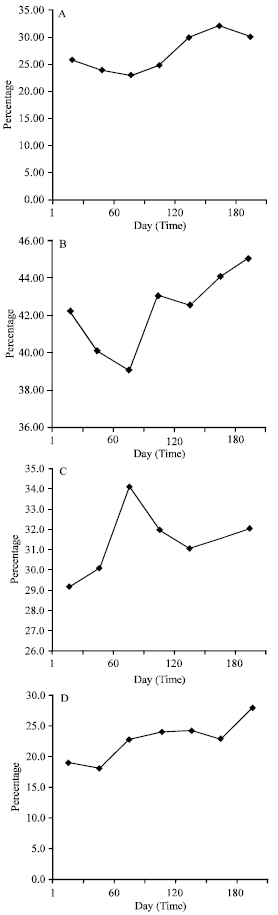 | |
| Fig. 1: | Change of sperm abnormality in four samples used data are average |
During sample observation (6 months) number of normal spermatozoa was fallen but there was not decrease significantly. Abnormal spermatozoa changes were illustrated in (Fig. 1).
Different shapes including; headless, tailless, middle part with tail, bent mid piece, shoe hook tail were observed. Abnormal spermatozoa were 30.83%. Shoe hook tail less than 2%. Changes in normal shapes showed downward trend, but it was not significant using t-test (5%).
Shape of spermatozoa is one of the important factor on fertility. Any kind of abnormalities in sperm’s shape disturbs its progressive motility and avoids penetrating to Zona Pellucida (ZP) of ovum. Figure 2 illustrates normal ram spermatozoa and its motile type and Fig. 3 illustrated different types of abnormal spermatozoa. Presence of protoplasmic droplets and any kind of abnormality, lead to progressive circular movement of spermatozoa (Mohammadzadeh and Doronin, 2002). Common abnormalities in sperm related to abnormal shapes tail (Molnar et al., 2001) and middle part. Most of these abnormalities were related bent mid piece tails and shoe hook tail and coiled sperm (Holt, 1982) were less common. Due to this types (coiled and shoe hook)observed in early stages of test so seems to be primary abnormality.
Although culture type should be considered in occurrence of abnormalities, Sankai et al. (2001) showed temperature (+10°C) were the reason for hardness and fragility of cell wall which result in falling flexibility and sperm’s motility. In this study most abnormality were observed in tail. Probably these abnormalities related to temperature.
According to Fig. 3 (coiled spermatozoa), related to disturbance in cell wall and during entering to epididymis completely coiled. This shape is produced in middle segment of epididymis and accumulated at the terminal part. Completely coiled shape is pathogen which is due to join protoplasmic droplets (Holt, 1982).
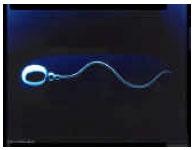 | |
| Fig. 2: | Normal shape of ram spermatozoa and type of motility |
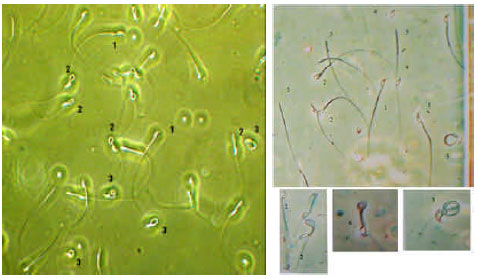 | |
| Fig. 3: | Different types of spermatozoa. 1) Normal 2) Bent mid piece 3) Coiled 4) Detached head 5) Mid piece and tail in mouse spermatozoa (Mohammadzadeh and Doronin, 2005) |
Losing, breaking or joining groups which stretch from middle part to tail causes producing bent mid piece spermatozoa (Lobl and Methews et al., 1978). Even coiled or broken tail was seen in samples but proportion of coiled and shoe hook tail was very low. Normal ram spermatozoa was resistant at +4°C and their quantitative changes was very low and most abnormalities were related to bent mid piece spermatozoa. These finding was shown proportion of coiled spermatozoa in ram semen is very low (2%) comprise to mouse semen and during to storing of ram semen at +4°C, normal spermatozoa don’t change to fragment (Detached head, mid piece or tail etc) but in mouse spermatozoa almost 20 days after storing at +4°C normal spermatozoa significantly was changed and decreased. Proportion of fragment increased (Mohammadzadeh and Doronin, 2005). According to Barrios et al. (2000) cell membrane of ram and bull spermatozoa is specific comprise somatic cells. So ram semen has high resistance and need to investigate cell integrity and mitochondria activity for determination sperm viability.
Thanks to Research Division of Lorestan University for financial and providing budget. Thanks to Mr. Seyed Mohsen Musavi and Dr. Manzar Hossein Akbar for cooperation.