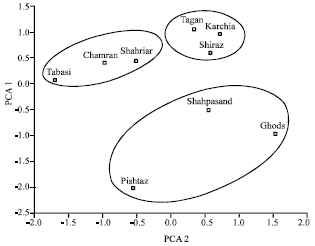
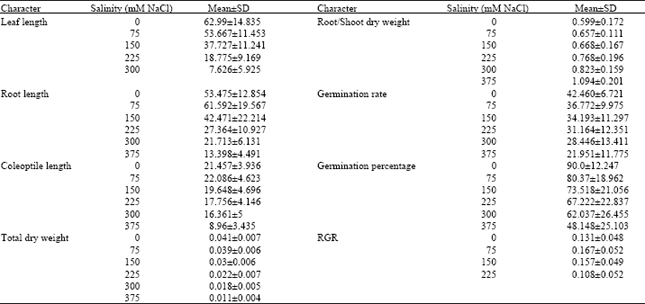
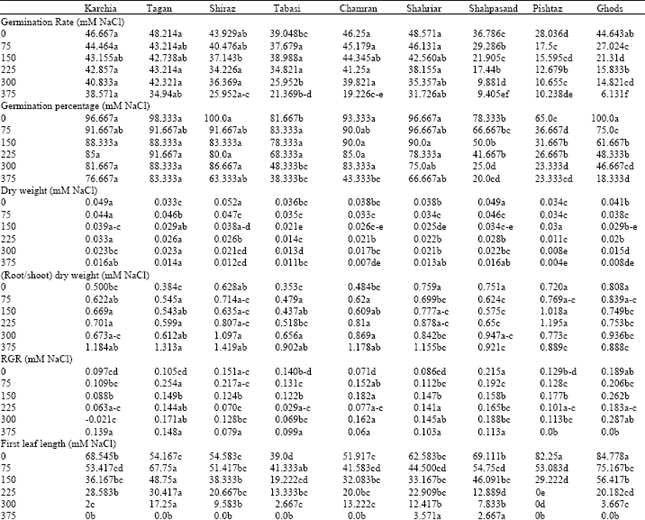
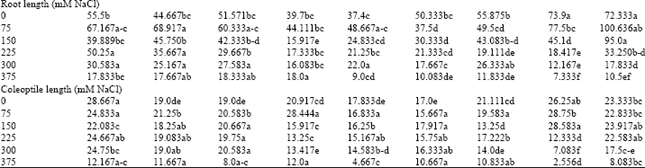
Research Article
Salinity (NaCl) Tolerance of Wheat Genotypes at Germination and Early Seedling Growth
Department of Biology, Faculty of Science, Azzahra University, Tehran, Iran
Khadijeh Kiarostami
Department of Biology, Faculty of Science, Azzahra University, Tehran, Iran
Fatemeh Behroozbayati
Department of Biology, Faculty of Science, Azzahra University, Tehran, Iran
Shokoofeh Hajihashemi
Department of Biology, Faculty of Science, Azzahra University, Tehran, Iran