ABSTRACT
The study of micro architecture of cancellous bone is important factor in diagnosis of bone diseases such as osteoporosis. Bone mineral density depends on factors like sex, age, genetic and environment. Bone mass, number of trabeculae, trabecular thickness decrease with aging in both sexes. The aim of this study was to investigate trabecular bone architecture in young healthy men and women. Iliac crest bone biopsies were taken from 53 healthy Caucasian men and women (28 men and 28 women, ages 20-49). The histomorphometry of cancellous bone was compared in undecalcified sections of iliac crest bone biopsies. Sections (8 μm) were analyzed by an automated trabecular analysis system (TAS) which measures a comprehensive range of structural variables such as bone volume, trabecular separation, number, connectivity and width. Although variables like bone volume, bone surface and trabecular thickness in men were more than women but this difference was not significant. Trabecular separation in men was less than women and this was significant. In addition number of trabecular nodes and index of node to terminus in men was more than women which this difference was not significant. Trabecular length was more in men than women and this was significant. In conclusion despite no significant difference in some of the above variables there is more mechanical strength due to less separation and more trabecular length in trabecular bone architecture in young men in comparison with young women.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2006.1338.1341
URL: https://scialert.net/abstract/?doi=pjbs.2006.1338.1341
INTRODUCTION
Total body bone mass changes with age. Bone volume reaches a maximum about 10 years after linear growth stops, probably begins to decrease somewhere in the fourth decade, and declines to half its maximum value by the age of 80 (Smith et al., 1975). Peak bone mass, which is attained in early adult life is dependent primarily on genetic factors (Mundy et al., 1994; 1995) and more recent studies measuring a bone mass at the spine and hip report peak value to be achieved during the 3rd and 4th decades (Lu et al., 2000). In another study the vertebral and femoral neck size and BMD (bone mineral density) was steeper with age in Caucasian boys than girls (Henry et al., 2004). Bone mass is lower in women than it is in men, and lower in Caucasians than in African-Americans (Riggs et al., 1981).
Women of all ethnic groups show an additional accelerated phase of bone loss that occurs for about 10 years after the menopause (Riggs et al., 1981). It has been estimated that a women can expect to lose 35% of her cortical bone and 50% of her cancellous bone as she ages, and a man can expect to lose about two-thirds of these amounts (Mazess, 1982).
Although cancellous bone comprises only 15% of the skeleton, the changes that occur in this type of bone after 30 determine whether the clinical features of osteoporosis will occur. The loss of cancellous bone that occurs with aging is not due simply to generalized thinning of the bone tra-beculae, but is rather due to complete perforation and fragmentation of trabeculae (Parfitt et al., 1983; Kleerekoper et al., 1985). Depending on the technique used, decline in cancellous bone mass begins in early adult life, occurring earlier than the decline in cortical bone mass (Riggs et al., 1986). Other studies have disagreed with these findings, and suggested that the decline in cancellous bone mass begins later, after ovarian function ceases (Genants et al., 1982).
In recent years different new techniques has been applied for measuring bone mass as a quantitative analysis of different parts of body in different sexes (Henry et al., 2004). Mechanical strength of bone is not just related to the amount of bone mineral density (BMD) and complex micro architecture of cancellous bone is a crucial factor in mechanical strength as well (Shahtaheri et al., 1999; Saino et al., 2003). However histomorphometry of trabecular bone is essential and the aim of this study was to compare micro architecture of trabecular bone in young men and women.
MATERIALS AND METHODS
The iliac crest bone biopsy were from 53 healthy Caucasian men and women (28 men and 28 women, ages (20-49) who died suddenly with no history of bone disease and no treatment likely to effect the bone.
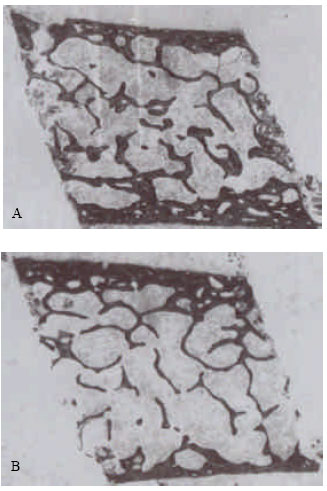 | |
| Fig. 1: | Photomicrographs showing the main transiliac histological features in (A) men and (B) women (x13) |
Specimens were fixed in 70% ethanol and embedded undecalcified in methyl methacrylate (Shahtaheri et al., 1999). Six sections, 8 μm thick were cut from each on a Jung K heavy duty microtome and stained by the 0.1% toluidine blue, pH 3.5 (Fig. 1). Since the sections were to be analysed using an automated image analyzing system it was essential that the stains chosen provided good contrast between bony tissue and the marrow spaces, otherwise their separation when tresholding to create a binary image would not be reliable. Each section was placed upon the stage of a low power microscope fitted with a zoom lens. After calibrating the system the image of the whole section was captured by a closed circuit television camera attached to the microscope and to a VIP image analyzer (Sight system, Newbury, UK). The captured image was threshold to separate the bone trabeculae from any background noise caused by the presence of stained marrow tissue. A binary image was created made up of 256 x 256 black and white pixels (Fig. 2).
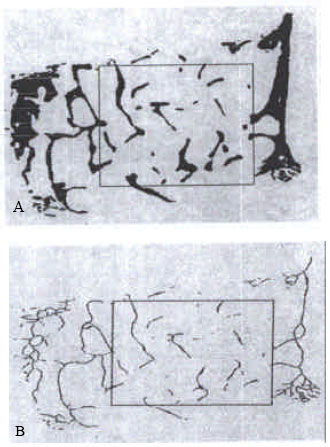 | |
| Fig. 2: | (A) Computer-generated printout showing the image of bone biopsy with inner and outer cortices and the trabecular region for analysis within a rectangular window. (B) The skeletonized image within the rectangular window ready for analysis |
Analysis consisted of: the measurement of intact image when the area of interest was defined by an elastic window followed by the measurement of the thinned image when the selected part of the binary image was skeletonized or thinned to its medial axis. The comprehensive range of variables measured such as bone volume, trabecular thickness and trabecular separation for intact image and node number (junction between 2 trabeculae), terminus number (free end of trabeculae) and the ratio of node to terminus as an index of connectivity for skeletonized image.
Statistical analysis: Results were expressed as the mean±SD or mean±SE and statistical significance of any differences between the 2 groups was determined using the Minitab software package and the Minitab t-test.
RESULTS
All of the measurements performed by TAS would be too cumbersome so the results were confined to nine of principal trabecular microanatomical variables. These are shown in Table 1 and include the trabecular bone volume, the bone surface, the trabecular thickness, separation, node number, terminus number, node/terminus ratio, total strut number and total strut length.
| Table 1: | Comparison of the trabecular microanatomy in Iliac crest bone biopsy of young men and women |
 | |
The bone volume was higher in young men than young women (p = 0.058). Bone surface was higher in young men than young women (p = 0.075). Trabecular thickness was higher in young men than young women (p = 0.6). However all of the above variables failed to reach significant. Trabecular separation (distance between trabeculae) was significantly lower in young men than young women (p = 0.03). Node number was higher in young men than young women (p = 0.05). However the terminus number and node terminus ratio as an index of connectivity were higher in young men compared to young women, the difference was not significant (p = 0.43). Total strut number was higher in young men than young women (p = 0.4) and trabecular length was significantly higher in young men than young women (p = 0.038).
DISCUSSION
The evidence showed that most histomorphometric variables were higher in young men than young women. Bone development and growth are similar in boys and girls up to the start of puberty. Thereafter, skeletal dimorphism evolves with a greater bone mass in adult males than in adult females (Vanderschueren et al., 2004). In current study there were more bone volume and bone surface in young men than young women which this supports earlier work suggesting lower bone volume in women than men (Smith et al., 1975; Riggs et al., 1981; Mazess, 1982). In another study reported men have more bone volume as a result of higher periosteal bone formation rates (Seeman, 2002).
In this study we also found that the trabecular connectivity in iliac crest bone biopsy was higher in young men than young women which are also consistent with previous studies (Thomsen et al., 2001). One of our major finding in this study was the less separation of trabeculae in young men than young women which support previous study (Thomsen et al., 2001).
 | |
| Fig. 3: | Mechanism of loss of trabecular bone in women and trabecular thinning in men |
In this study we found trabecular length and number were higher in young women than women which is consistent with previous studies via histomorphometric techniques (Thomsen et al., 2001). Also, there is a report which explained the amount of trabecular bone lost during aging is less in men than in women (Mosekilde, 1990) which supports above results. In later life the loss of trabecular bone in men proceeds in a linear fashion with thinning of trabeculae rather complete loss, as is seen in women (Aaron et al., 1987). Bone loss is the result of a reduction in the volume of bone removed in the basic multicellular units, so trabecular connectivity and less separation between trabecular connectivity is better maintained in men than women (Fig. 3). However, trabecular bone loss occurs mainly by thinning in men and mainly by loss of connectivity in women (Aaron et al., 1987). In women after menopause, menopause-related oestrogen deficiency increases remodeling and at each remodeled site more bone resorbed and less is formed, accelerating bone loss and causing trabecular perforation and disconnection (Seeman, 2002). More women than men sustain fractures because their smaller skeleton incurs greater architectural damage and adapts less effectively by periosteal bone formation. In conclusion cancellous bone microarchitecture has more microarchitectural strength in young men than young women presented more mechanical strength in cancellous bone microarchitecture in young men than young women.
ACKNOWLEDGMENTS
I would like to thank for great support of Dr. Jean Aaron and Mrs. Pat Shore at the School of Biomedical Sciences, University of Leeds, England.
REFERENCES
- Henry, Y.M., D. Fatayerji and R. Eastell, 2004. Attainment of peak bone mass at the lumbar spine, femoral neck and radius in men and women relative contributions of bone size and volumetric bone mineral density. Osteopos Int., 15: 263-273.
Direct Link - Kleerekoper, M. and A.R. Villanueva, 1985. The role of three dimensional trabecular microstructure in the pathogenesis of vertebral compression fractures. Calcified Tissue Inl., 37: 594-597.
Direct Link - Shahtaheri, S.M., J.E. Aaron, D.R. Johnson and S. K. Paxton, 1999. The impact of mammalian reproduction on cancellous bone architecture. J. Anatomy, 194: 407-421.
Direct Link








