Research Article
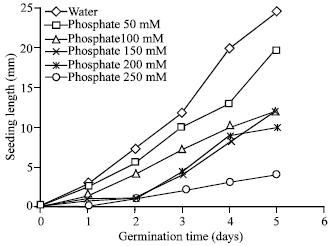
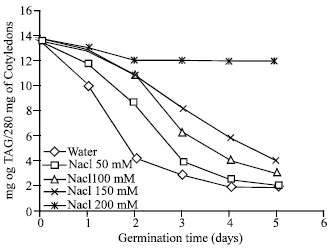
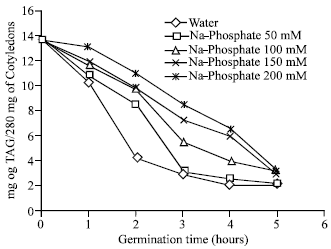
Ionic Effect on Mobilization of Seed Storage Nutrient Substances and Lipase Activity in Germinating Oil Seeds (Brassica napus L.)
Department of Biochemistry and Molecular Biology, University of Rajshahi, Rajshahi-6205, Bangladesh
N. K. Sana
Department of Biochemistry and Molecular Biology, University of Rajshahi, Rajshahi-6205, Bangladesh
I. Hossain
Department of Biochemistry and Molecular Biology, University of Rajshahi, Rajshahi-6205, Bangladesh