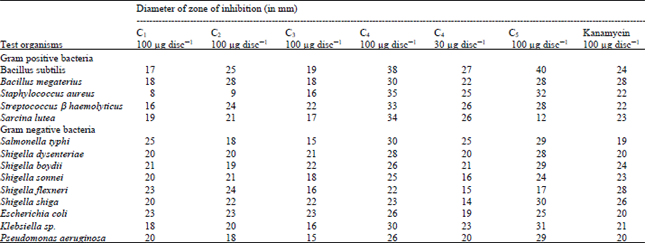
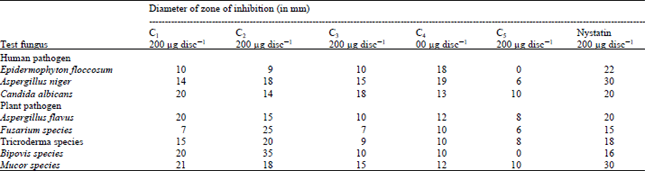
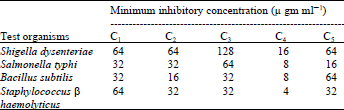
Research Article
In vitro Antimicrobial Screening of Three Cadmium Coordination Complexes and Two Addition Compounds of Antimony and Arsenic
Department of Chemistry, Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
M. Aziz Abdur Rahman
Department of Chemistry, Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
M.A. A. Al-Bari
Department of Chemistry, Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
M.L. A. Banu
Department of Chemistry, Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
M. Saidul Islam
Department of Chemistry, Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
N. A. Khatune
Department of Chemistry, Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
Golam Sadik
Department of Chemistry, Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh