Research Article
Role of Nitrate Reductase for N Assimilation During the Grain Filling Period of Been Seeds (Lablab purureus L.)
Department of Biochemistry & Molecular Biology, University of Rajshahi, Rajshahi - 6205, Bangladesh
Bean is a good source of proteins and minerals which helps in the maintenance of proper health and resistance to disease. The seeds of bean occupy the leading position as a pulse food in Bangladesh. In several districts of Bangladesh and some other countries of this subcontinent the splinted bean seeds are used as " dhal" called " seim dhal". The seeds contain, water 9.6%, protein 24.9%, fat 0.08-1.1%, carbohydrate 50.1% and mineral matter 4% (Purse Glove, 1969). The protein of beans are similar to those of lentils and peas Protein yield was increased by soil or foliar fertilization with N late in the growing season (Finney et al., 1957; Hucklesby et al., 1971). This suggests that an insufficient supply of N to the kernels during grain filling stage limited protein production in control plants in these studies. Nitrogen used in grain protein synthesis is either remobilized from the vegetative tissues where it was accumulated before anthesis, or is newly assimilated from the soil after anthesis. Although both processes may occur simultaneously, the capacity of vegetative tissues to continue to assimilate N during the grain filling period likely becomes increasingly limited as those tissues undergo senescence. It might expect the contribution to the grain of N newly assimilated after anthesis to be low. The purpose of this research work was to begin to identify and refine the understanding of those factors limiting N assimilation after anthesis.
Recently, the Genetics and Breeding Department of Rajshahi University cultivated many cultivars of bean by cross breeding. For our investigations we used seeds of three different cultivars of bean such as Dolichos, Ipsha-1, and Ipsha-2. In this study relationship was characterized between in vivo nitrate reductase (NR) enzyme activity and post anthesis N accumulation for three tub-grown cultivars. In addition, using a non nitrate modification of the in vivo NR assay, the importance of nitrate availability was evaluated during the post anthesis period.
Germination and collection of seeds: For experimental purposes the mature seeds of different cultivars of bean (Dolichos, Ipsha-1 and Ipsha-2), were collected since 1999 to 2000 from the Department of Genetics & Breeding, Rajshahi University, Rajshahi.. Bangladesh. Good and mature seeds of bean were soaked in distilled water within a glass beaker for 6 h. A random lot of 20 been seeds of new cultivars were sown at the mid July in each earthen tub (20 inches dia.) containing soil, homogeneously mixed with local fertilizer (Cow dung). Besides that, 2 kg/100 tub NO3 -N; Nitrogen (5.5 kg/100 tub), P (2.5 kg/100 tub) and K (4.5 kg/100 tub) were incorporated into the soil before planting. The fertilizer was supplied in two applications; the last one applied before two weeks of harvesting. The plants were grown in open place (on the roof) and were watered daily. A later application of ammonium nitrate (5.5 kg N/100 tub) was made on 20th August. Grasses were, however, pulled up time to time. Anthesis occurred for all three cultivars on 22nd to 28th September. The grain filling periods for all cultivars were nearly the same.
Collection of leaves: The flag leaf blade, flag leaf sheath and the first leaf blade below the flag leaf (penultimate leaf) were assayed for NR activity twice weekly after anthesis. These tissues possessed the highest levels of NR activity during much of the post-anthesis period and accounted for approximately 50-70% of the NR activity in shoots during this time.
Five shoots of each cultivar were sampled at random from each of three replicates on each date. These were transported to the Laboratory on ice and the tissues noted above were removed. Like tissues were bulked for each replicate and weighed. Leaves were split along the mid-rib and sliced into small sections which were used in the assays. Sheaths were also prepared for the assays by cutting into small sections.
Measurement of NR activity : Nitrate reductase activity in the tissues was estimated by an in vivo assay based on the method of Hageman et al. (1971). A no-nitrate modification, as proposed by Radin et al. (1975), was also used. Duplicate 0.3 g samples of the plant parts from each replicate were placed in 50 ml Erlenmeyer flasks containing 5 ml of ice-cold infiltration medium containing 0.1 M phosphate buffer (pH 7.5) and either no nitrate (digested no nitrate method) or 0.05 M nitrate (digested + nitrate). It was found that 0.05 M nitrate gave the maximum activity in these tissues. Tergitol (0.2% v/v), as a surfactant, increased the observed NR activity and was included in both mediums. The samples were vacuum infiltrated four times (2 min each). The flasks were flushed with N2 gas, stoppered immediately and incubated in the dark at 30 °C. Those flasks with nitrate in the medium were incubated for 1 hour. Those without nitrate were incubated 30 min since it was determined that nitrate production might become nonlinear after that time. A 0.2 ml aliquot taken after the incubation period was used to measure nitrate efflux from the tissue during incubation.
Measurement of nitrate: Nitrate was measured colorimetrically as described by Hageman et al. (1971) except that sulfanilamide reagent was 1% (w/v) in 3 N HCl. Nitrate reduction per shoot during the grain filling period was predicted from the total NR activity of the flag leaf blade, flag leaf sheath and penultimate leaf blade combined. The NR activities detected on two successive sampling dates were averaged and the resulting mean activity was used to calculate the quantity of nitrate potentially reduced by these tissues for each day in the period among the three samples. A diurnal variability factor of 0.75 suggested for wheat by Dalling et al. (1975), was used. No attempt was made to account for nitrate potentially reduced by tissues other than those assayed.
N estimated by kjeldahl method: To estimate the quantity of reduced N actually accumulated by the cultivars, all above-ground vegetation was sampled from each of four replicates for each cultivars on 11September (1 day after anthesis), 24 Sept. 10 Oct. and 18 Nov. (ripe grain). Four of these replicates were the same tub from which the NR activity samples were obtained. The number of shoots in each sample was counted. Samples were dried for 48 h at 65°C, weighed and analyzed for N by a micro-Kjeldahl procedure. Reduced N content was expressed on a per shoot basis by Wong (1923).
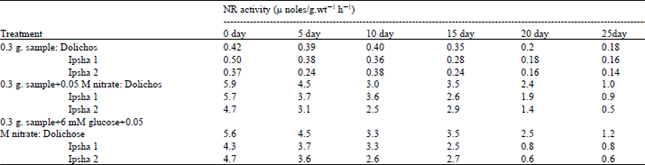
Experimental results from three cultivars of bean seeds showed minimal accumulation of reduced N after anthesis, which agrees with other field of observations for wheat (Eilrich et al., 1973; Austin et al., 1975). The flag leaf blade possessed highest NR activity during most of the sampling period, followed in order by the penultimate the leaf blade and flag leaf sheath (Figs. 1A-C ). The decline in activity over the period appeared to parallel the senescence of these tissues, estimated visually. The penultimate leaf senesced earliest, followed by the flag leaf blade and finally, the flag leaf sheath. All three cultivars displayed similar trends in activity over the period with Dolichos tending toward higher activity, although in most cases not significantly so.
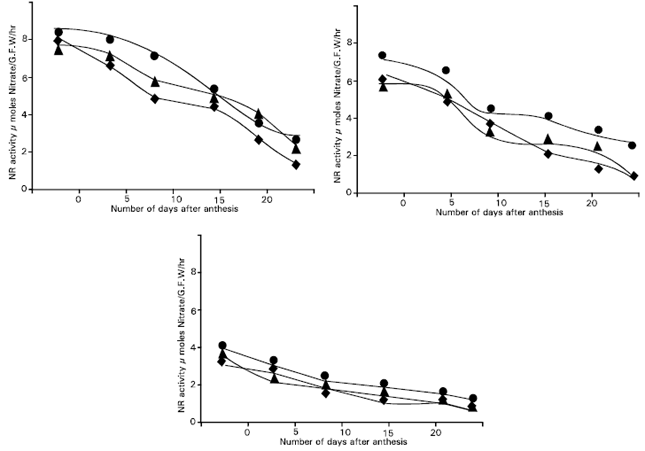
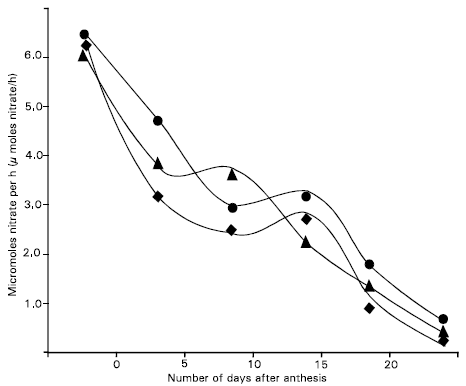
The in vivo assay modification in which no nitrate was included in the infiltration medium gave only low level of NR activity compared with the + nitrate method (Fig. 2 and 3). This activity showed little change over the sampling period in contrast with declining trend observed with the + nitrate procedure. One interpretation of these results is that the enzyme in these tissues was not being fully utilized, particularly at the earlier dates of the sampling period. Although nitrate levels in these tissues were not measured, the no-nitrate assay results suggest that little nitrate was available in these tissues for reductions. The tissues did seem to have substantial levels of the nitrate-induced NR enzyme. One explanation for this conflict may be that there are two separate pools of nitrate in the tissues, one concerned with regulation of the level of NR and the other with the supply of nitrate as substrate (Dalling et al., 1975).
| Table 1: | Effect of (+ nitrate) and (+ nitrate and + glucose) on the NR activity in been leaves (Dolichos, Ipsha 1, Ipsha 2 variety) |
 | |
 | |
| Fig. 1: | Nitrate reductase activity during the grain filling period in the (A) flag leaf blade, (B) flag leaf sheath and © penultumate leaf blade for ● Dolichose, ▲ Ipsha-1 and ◆ Ipsha-2 varieties of bean, assayed by the +notrate assay modification |
Glucose has been shown to enhance the NR activity observed in morning glory and maize leaves (Klepper et al., 1971). It was proposed that added glucose increased the availability of NADH to NR enzyme. Inoculation of glucose (6 mM) in the + nitrate assay in our experiment (Table 1) did not consistently alter the activity detected. This suggests that availability of NADH was not a primary factor limiting the nitrate reduction in these tissues.
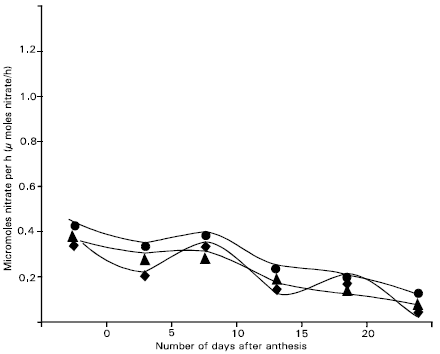
Reduced N accumulation by a single shoot after anthesis was predicted from the combined NR activities of the flag leaf, penultimate leaf and flag sheath, (Fig. 4). In actual reduced N accumulation measured on samples from the same plot area is also shown. When NR activity estimated by + nitrate assay was considered, it appeared to be potential for considerably more reduced N accumulation than was actually observed.
Croy and Hageman (1970) proposed that low NR activity in winter wheat late in the growing season limits protein production by the grain.
 | |
| Fig. 2: | Total nitrate reductase activity for Dolichos, Ipsha-1 and Ipsha-2 in the flag leaf blade, flag leaf sheath and penultimate leaf blade combined as measured by the + nitrate assay modification. (● Dolichos, ▲ Ipsha-1 and ◆ Ipsha-2) |
 | |
| Fig. 3: | Total nitrate reductase activity for Dolichos, Ipsha-1 and Ipsha-2 in the flag leaf blade, flag leaf sheath and penultimate leaf blade combined as measured by the no nitrate assay modification. (● Dolichos, ▲ Ipsha-1 and ◆ Ipsha-2) |
Experimental data would suggest that Dolichos, Ipsha-1 and Ipsha-2, during much of the sampling period, substantial level of NR enzyme were present in the tissues but that insufficient nitrate was being supplied to the enzyme. Chantarotwong et al. (1976) concluded that reduction of nitrate in barley seedlings was more the function of rate of nitrate uptake by the plant than of the level of NR activity (in vitro measurement).
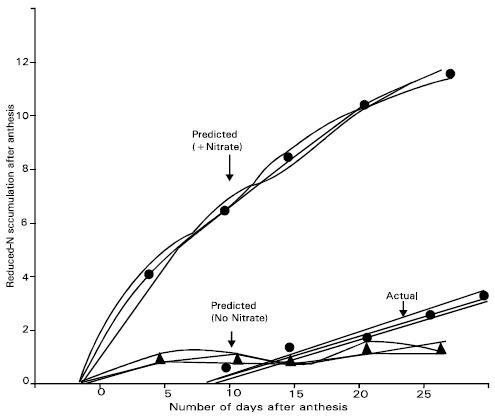 | |
| Fig. 4: | Actual and perdicted reduced N accumulation per shoot after anthesis for Dolichos, Ipsha-1 and Ipsha-2 considered together. Prediction were calculated using nitrate reductase activities of the flag leaf blade, flag leaf cheath and penultimate leaf blade combined, as measured by both the + nitrate and no-nitrate assay modifications |
Rao et al. (1977) also found that non specified factors permitted only a part of the potential NR capacity of their wheat genotype to be utilized.
Efforts to enhance post-anthesis N assimilation in spring wheat may therefore need to be directed at an enzyme or process other than NR. Such a process might be the actual uptake of nitrate from the soil or its subsequent transport to the site of reduction. Considerable attention has been given to senescence of shoot tissue after anthesis. Perhaps more consideration should also be given to senescence of the root system and the associated decline in those processes in the roots involved with N assimilation. We do not propose that the level of nitrate reductase enzyme is never a limiting factor. During vegetative growth or perhaps even towards the end of grain filling when NR activities have declined to low levels, nitrate reductase might be an important consideration. However, present results suggested that the minimal accumulation of reduced N after anthesis often noted in wheat is not primarily the result of limiting levels of nitrate reductase enzyme.