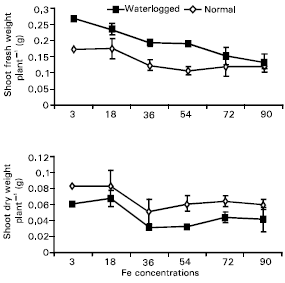
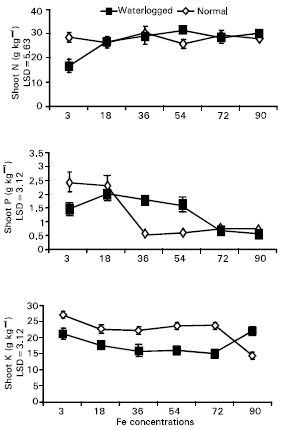
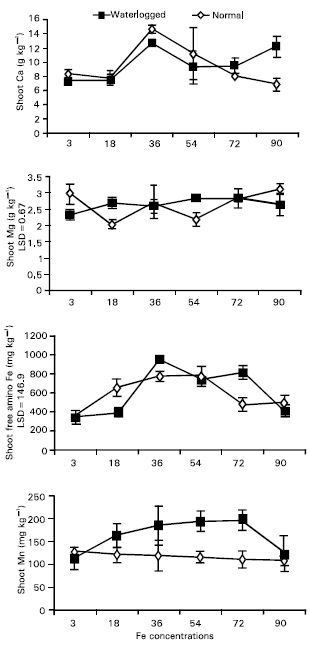
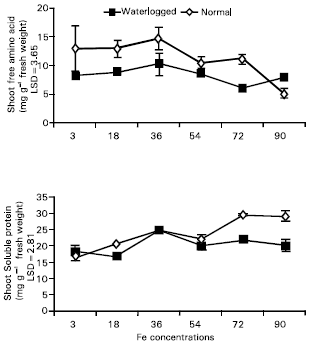
Research Article
Water Logging Tolerance in Populations of Cynodon dactylon from Contrasting Habitats
Institute of Pure and Applied Biology, Bahauddin Zakariya University, Mullan, Pakistan
Seema Mahmood
Institute of Pure and Applied Biology, Bahauddin Zakariya University, Mullan, Pakistan
Rubina Shaheen
Institute of Pure and Applied Biology, Bahauddin Zakariya University, Mullan, Pakistan
Muhammad Ashraf
Department of Botany, University of Agriculture, Faisalabad, Pakistan