Research Article
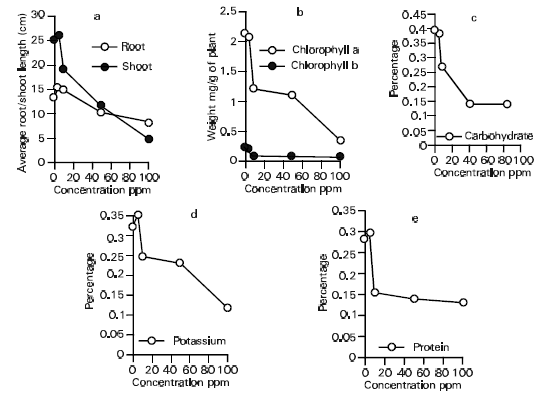
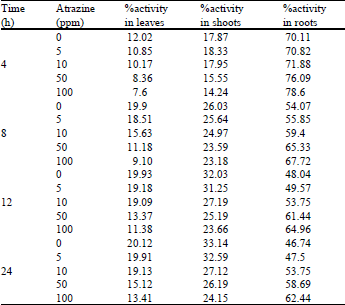
Effect of Atrazine on Absorption and Translocation of P32, Chlorophyll, Carbohydrate, Protein and Potassium Contents in Bean Vigna radiata (L.) Wilczek
PCSIR Laboratorie Complex Off. University Road, Karachi-75280, Pakistan,
Rashida Perveen
Department of Chemistry, University of Karachi, Karachi�25270, Pakistan
Iftikhar Imam Naqvi
Department of Chemistry, University of Karachi, Karachi�25270, Pakistan
S. Shahid Shaukat
Department of Botany, University of Karachi, Karachi�25270, Pakistan