ABSTRACT
Biological and pharmacological assays of the extracts of Gentiana oliveri and its pure alkaloid gentianine indicated to be highly efficacious. Toxicity in vertebrates was evaluated by chronic toxicity assay in rats. LD50 was found to be greater than 1000. Slightly elevated values of serum glutamate pyruvate transaminase and lactate dehydrogenase were observed at higher doses, but hypoglycemia did not occurred. At lower doses it was found to be non-toxic.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2003.1949.1950
URL: https://scialert.net/abstract/?doi=pjbs.2003.1949.1950
INTRODUCTION
The plant of genus Gentiana has been the subject of interest from medicinal point of view. G. olivieri is locally used from centuries as a remedy for many diseases (Perry, 1980). Previous workers have reported the presence of alkaloids (Rakhmatullaev and Yunusov, 1972) in this plant but no significant biological research was conducted. In our series of publications we have reported its remarkable hypotensive effect (Mansoor et al.,1998); antibacterial and antifungal effect (Mansoor et al., 1999) and non-cytotoxic effect (Mansoor et al., 2000). These important properties of G. olivieri must be utilized, by developing it into an efficacious drug, so that the people could be benefited by its therapeutic effects. However, the adverse effects or the side-effects of the drugs should be known before using it as a medicine. All drug regulatory agencies require that any drug that is to reach the market should be subjected to rigorous evaluation to determine its safety and efficacy.
The present paper describes the work done to find out the toxic levels and other side-effects of aqueous extract of G.olivieri and gentianine on rats.
MATERIALS AND METHODS
The plant material: The fresh plant material of Gentiana olivieri was collected from Chiltan heights near Quetta. After drying and grinding, crude ethanolic extract was prepared and its alkaloid gentianine was purified by the method described by Mansoor et al. (2000).
Chronic toxicity assay: Thirty-six male wistar rats weighing 200-250 gm were selected and placed in six groups of six rats each. All the rats were kept on their routine diet and water in the metabolic cages. Increasing dose of 200-600 mg kg-1 gentianine and aqueous extract of G.oliveri were given to each group separately. One group of rats was kept as a control. Gentianine and the aqueous extract were dosed daily, orally, with the help of a syringe till seven days. On the seventh day, 3-4 hours after feeding the animals were weighed and then killed by chopping off their heads. Blood was collected separately to perform biochemical tests. Urine and stool were also collected during the week for physical examination (Saxena et al., 1993).
Parameters of analysis: The biochemical blood tests performed for each sample were, the determination of glucose concentration, lactate dehydrogenase (LDH) concentration and serum glutamate pyruvate transaminase (SGPT) concentration. Glucose, LDH and SGPT were determined by kit methods, provided by Merck.
RESULTS AND DISCUSSION
Table 1 indicates that the aqueous extract of G.olivieri and gentianine do not have any toxic effects on the rats. All the rats survived even at higher doses of the pure compounds and aqueous extract, although administered for a long period of seven days. Slight decrease in the weight of the rats was recorded for those who were fed with the aqueous extract. This loss might be due to the bitter taste of extract (Ahmad, 1994). The LD50 estimated was >1000 which indicates that the drug is not lethal and could safely be given to the vertebrates. The LD50 test provides a gross indication of the overall toxicity of the drug (Rogers and Spector, 1986).
| Table 1: | Levels of dose administration of aqueous extract and Gentianine in rats |
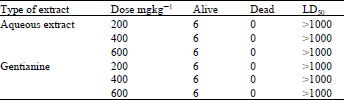 | |
| LD50: Lethal concentration causing death for 50% population. | |
| Table 2: | Mean ± SEM blood values of glucose, serum glutamate pyruvate transaminase (SGPT) and lactate dehydrogenase (LDH) of Controls and after oral administration of gentianine and aqueous extract of G. olivieri |
 | |
There was no change in the physical appearance of the stool. Urine volume of the treated rats was in much more quantity as compared with the controls. This might be due to the diuretic property of Gentiana species as appeared in the literature (Perry, 1980).
During the behavioral observation, the major signs observed were decreased activity and passivity especially to the rats given the higher doses of both Gentianine and the aqueous extract. The decrease in activity started on the third day of the dose. This could be due to excessive urination, resulting in decreased blood pressure (Rodriguez et al., 1994) and general weakness. Further studies of its effect on blood pressure and excessive urinary output must be carried out, so as to develop this alkaloid as a hypotensive agent.
Blood sampling for biochemical evaluation revealed interesting results. Mean values of blood biochemical analysis of the treated and the control group are given in Table 2. The groups of rats treated with aqueous extract, indicate slight, negligible, decrease in the blood glucose level as compared with the control. This could be attributed to the bitter taste of the extract, causing lesser intake of food. Thus, passivity of the rats after chronic alkaloid administration was not due to hypoglycemia. SGPT levels were slightly affected by gentianine. Its concentration rose comparatively to a greater extent in the rats dosed with aqueous extract. LDH concentrations appear to be most effected by both the samples, especially at higher concentrations. Levels of LDH appear to increase in a dose dependent manner. Both LDH and SGPT are very important body enzymes and their elevated values could be toxic to the body.
Thus, higher doses of gentianine and crude extract of G. olivieri are of low toxicity to the body and should therefore be avoided.
REFERENCES
- Rakhmatullaev, T.U. and S.Y. Yunusov,, 1972. Alkaloids of Gentiana olivieri. Chem. Natl. Compounds, 8: 344-345.
CrossRefDirect Link








