Research Article
Role of Light in the Regulation of the Nitrate Reductase level in Lentil (Lens esculenta L.)
Department of Biochemistry and Molecular Biology, University of Rajshahi, Rajshahi-6205, Bangladesh
The exact role of light in controlling NR activity has not yet been established. Some of the researcher (Jones and Sheard 1975 and Tompkins et al., 1978) could not found , a strict correlation between the level of NR and levels of endogenous nitrate. But Beevers et al. (1972), proposed that light might act for making higher concentrations of nitrate within the cell. Sawhney and Naik (1972) suggested that the redox changes in greening tissues, as a result of Hill reaction, create favorable conditions for the enhanced rate of synthesis of the enzyme. Travis and Key (1971) suggested that the light induced increase in polysome formation produces a nonspecific effect on the rate of NR synthesis. Later Aslam et al. (1976) showed that light affects the movement of nitrate from storage to the metabolic pool. Another suggestion by Alsam et al. (1973), however could induce NR in carbohydrate sufficient plants even in the dark and concluded that respiration is more important for the induction process. Many researcher (Jones and Sheard, 1972; Duke and Duke, 1978 and Johnson, 1976) demonstrated that Phytochrome controls the NR level in pea terminal buds.
The present investigation was carried out with excised leaves of Lentil (Lens esculenta L.) to determine the way of light acts in controlling NR activity. Attempts were made to find out if the light effect is mediated via photosynthetic or non-photosynthetic reactions. The time course of increase in NR activity modulated by light suggests de novo synthesis. Further, it was found that the enzyme level is controlled by the photoreceptor, Phytochrome. This suggests that a close correlation exists between the process of nitrate reduction and the control of NR synthesis.
Plant material and treatments: Lentil seeds (Lens esculenta L.) were soaked in water for 6 h, then germinated in petri-dishes in the dark at 25°C. Watering and all other operation were performed under a green safe light. Leaves were excised from 9 day old dark grown seedlings of uniform height. These were weighed (250 mg), then floated with their basal side down in 20 ml of the incubation medium, the composition of which and other conditions during treatment are describe in the respective tables and figures. The pH of the incubation medium was adjusted to 6.0 and the temperature during incubation was maintained at 25°C. All treatments were given in duplicate and each experiment was repeated at least twice.
Enzyme extraction and assay: Leaves were homogenized in 2 ml of the extraction buffer containing 50 mM potassium phosphate (pH 7.5), 1 mM EDTA and 1mM cysteine, in a mortar at 4°C. The resulting mixture was centrifuged at 6000 rpm for 15 min at 4°C. The supernatant obtained was used to assay NR activity following the procedure of Hageman et al. (1971). Nitrate reductase activity is expressed as the number of μ moles of nitrite formed g fr wt–1 h–1
Light sources: Green safe light was obtained by filtering light from a 40 W cool white fluorescent tube through 7 layers of green cellophane papers. The intensity of light at plant level was never more than 1 μW/ cm2 . Red and far -red lights were obtained as described in detail (Sharma et al., 1976). Blue light was obtained from two 100 W tungsten lamps wrapped with 2 layers of blue cellophane paper.
Nitrate measurements: Excised lentil leaves (10 g), floated on 100 mM potassium nitrate in light for 19 hr were homogenized in the same buffer (50 ml) used to extract the enzyme, then they were centrifuged at 10000 rpm for 15 min. After the supernatant had been passed through a Sephadex G-25 column, it was used to estimate nitrate in the desired sample.
As shown in Fig. 1, the NR activity induced in leaves in total darkness was very low as compared that induced in light. At the optimal concentration (100 mM) of nitrate, an about 14 -fold higher activity was induced in light than in the dark. The time course kinetic of induction in showed a lag phase of 2.5 h followed by a rapid increase up to 20 h, after which activity decreased (Fig. 2). The decline in activity was immediate and about 14% of the activity was lost within 24 h. This kinetics pattern is typical of induced enzyme synthesis.
For low activity in the dark , Travis et al., 1969; suggested that an enzyme synthesized in the dark inactivated NR. To determine the true in the present system, it was mixed the extract (1:1 ) from light and dark incubated leaves, then followed their in vitro decay rate. It was found that the decay of the mixed extract did not differ from that of leaves incubated in light as shown in Fig. 3. This showed that there may not be any inactivating enzyme synthesized in the dark in this system.
To determine if any inactivate NR was synthesized in the presence of nitrate in dark ( which is activated in light) it was transferred leaves that had been incubated first in the dark with nitrate for 19 h to light after an addition of tungstate. Table 1 shows that tungstate completely inhabited the development of NR activity, whereas in the control slow induction seemed to continue up to 3 h after which activity increased to 6.1 enzyme unites at 10 h. [one enzyme unite being equal to 1 μ mole of nitrate formed g fr wt–1 h–1]. So it was inferred that no inactive NR synthesized in dark.
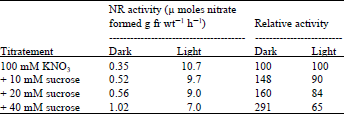
Effect of sucrose and DCMU on nitrate reductase induction: Sucrose is reported to enhance the NR level in several systems ( Higgins et al., 1974; Sluiters-Scholoten, 1975; Travis et al., 1970). In lentil leaves, sucrose added at 10, 20 and 40 mM concentration with 100 mM nitrate, did not enhance the NR level in light, in fact the levels decreased by 10 to 30% as compared to the control values with increasing concentrations of sucrose. However in the dark, with sucrose there was an increase in NR activity, which was 48 to 191% more than that of the dark control values.
| Table 1: | Effect of tungstate on the induction of nitrate reductase in lentil leaves that had been incubated first with nitrate in dark |
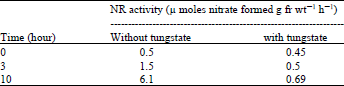 | |
Excised etiolated leaves were floated on 100 mM KNO3 for 19 h in the dark. Tungstate then was added (final concentration, 1 mM) while still in the dark and after half an hour the leaves were transferred to light. | |
| Table 2: | Effect of sucrose on the induction of nitrate reductase in lentil leaves |
 | |
| Table 3: | Effect of DCMU on the induction of nitrate reductase in lentil leaves |
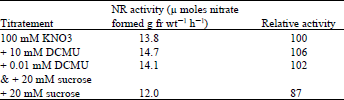 | |
| Table 4: | Effect of dinitrophenol and arsenate on nitrate reductase in lentil leaves |
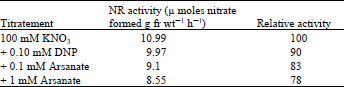 | |
| Table 5: | Effect of short period of light on the induction of nitrate reductase in lentil leaves |
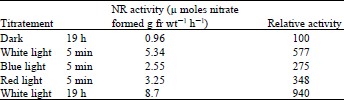 | |
The presence of even 40 mM concentration of sucrose in the dark, however, could not replace the light effect (Table 2).
Leaves excised from 9 day-old dark grown lentil seedlings were floated on 100 mM nitrate or 100 mM nitrate containing different concentrations of sucrose, then placed in light. NR activity was assayed after 19 h. To investigate the possibility of the direct involvement of photosynthesis in NR induction in light, the enzyme was induced in the presence of 0.01 mM ) DCMU [ 3-(3,4-dichlorophenyl)-1,1-dimethlurea] and 20 mM sucrose. Results in Table 3 show that DCMU failed to inhibit NR induction. With 20 mM sucrose there was a 13% decrease in activity but with DCMU the activity recovered.
| Table 6: | Control of nitrate reductase by Phytochrome in lentil leaves |
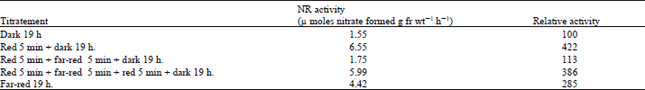 | |
| Table 7: | Measurement of the internal nitrate concentration in lentil leaves |
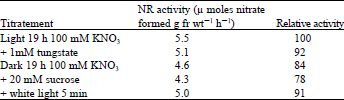 | |
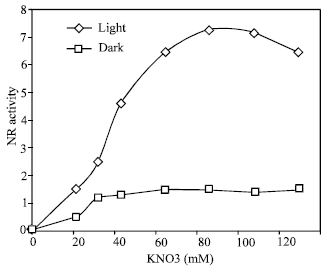 | |
| Fig. 1: | Dose response to potassium nitrate on nitrate reductase induction in light and in the dark. NR activity was measured after 19 h in light and in the dark. NR activity is expressed as the number of μmoles nitrate formed g fr wi1 h–1 |
Leaves from 9 day-old dark grown lentil seedlings were excised and floated on the specific treatment solutions, then placed in light. NR activity was assayed after 19 h.
Role of respiration in nitrate reductase induction: Since sucrose, as well as DCMU, did not affect NR induction in light, the oxidative phosphorylation uncouplers, DNP and arsenate, were added to the induction medium, and their effects studied to find out if respiration was driving the induction of NR as suggested by Aslam et al. (1973). As seen in Table 4, DNP and arsenate had only a slight effect on NR induction. DNP at a 0.1mM concentration inhibited NR induction by 10% and arsenate at 0.1 and 1 mM inhibited it by 17 and 22% respectively.
Leaves from 9 day-old dark grown lentil seedlings were excised and floated on the specific treatment solutions, then placed in light. NR activity was assayed after 19 h.
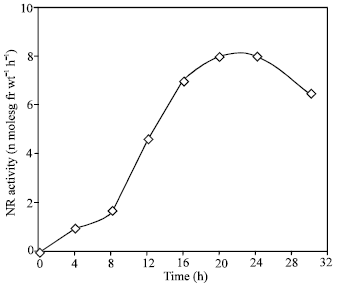 | |
| Fig. 2: | Time course study of nitrate reductase induction in the presence of potassium nitrate in light. NR activity is expressed as the number of n moles nitrate formed g fr wt–1 h–1 |
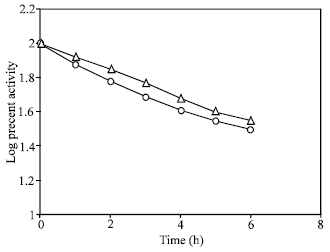 | |
| Fig. 3: | Effect of mixing enzyme extracts from light and dark induced leaves on nitrate reductase stability. In light induced leaves (- Δ-) 100% activity is equal to 9.5 μ moles nitrate formed g fr wt–1 h–1 ; in the mixed extract (-O-), 100% activity is equal to 5.8 μ moles nitrate formed g fr wt–1 h–1 |
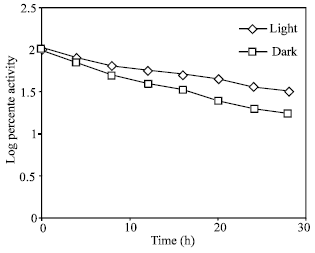 | |
| Fig. .4: | In vivo decay of nitrate reductase. The lose in activity was followed for 24 h in both light (-◊-) and in the dark (-□-). 100% activity is equal to 13.9 μ moles nitrate formed g fr wt–1 h–1 |
Influence of the short duration of different qualities of light and the involvement of Phytochrome: When exposed to white light for five minutes at the beginning of the induction period, the NR activity developed was 62% of that obtained after the leaves had been exposed to continuous white light for 19 h (Table 5). Similarly, expose to blue or red light for five minutes also enhanced NR activity; the range of enhancement by red light varied from 200 to 390% in five different experiments. These results and those obtained with DCMU suggest that photosynthesis is not directly involved in NR induction.
Excised leaves from 9 day-old dark grown wheat seedlings were floated on 100 mM potassium nitrate. The leaves exposed to white, blue and red light for 5 min, then kept in the dark for 19 h. Red light ( 500 mw cm–2) and blue light (700 mw cm–2) were obtained from two 100 w tungsten lamps wrapped with 2 layers of red cellophane paper and 2 layers of blue cellophane paper respectively. White light intensity was 1200 mw cm–2.
The induction of NR has been reported to be controlled by Phytochrome in mustard, pea and maize (Duke and Duke, 1978; Johnson, 1976; Jones and Sheard 1975). Since five minutes of red light irradiation increased the NR level in the present system, the possibility of Phytochrome involvement was also explored by testing the photo-reversibility of this response due to red and far-red lights. Leaves from etiolated seedlings were excised and floated on nitrate, after which they were given 5 min of red light and left for 19 h in dark. With an another set of leaves, 5 min of far-red light given immediately after the 5 min of red light. As shown in Table 6, red light increased the activity by 322% over that of the control; this was totally inhibited by subsequent exposure to far red light.
Excised etiolated leaves from 9 day-old dark grown lentil seedlings were floated on 100 mM potassium nitrate. The leaves exposed to the specified quality of light for 5 min, then kept in the dark. Enzyme activity was assayed after 19 h. Red light was obtained as described in Table 5. Far-red light was obtained from a 300 w tungsten lamp (USA) after filtering it through 8 cm of constantly flowing tap water and a CBS -750 FILTER . The intensity of the far-red light was 140 mw cm–2.
This phenomenon of photo-reversibility was observed even after far red light was followed again by red light. Also, in continuous far-red light the activity increased.
Effect of light on the intracellular nitrate level: According to some reports (Beevers et al., 1965) light enhances NR induction by facilitating nitrate entry. Therefore, to see if nitrate entry is blocked in the dark , we measured the intracellular nitrate concentrations. As seen in Table 7, nitrate did accumulate in the dark, the value being similar to that found in leaves exposed to light, though it was unable to induce NR.
Table 6 Control of nitrate reductase by Phytochrome in lentil leaves. If tungstate, which inhibits the development of active NR, is included in the induction medium, the nitrate accumulation is comparable to that of the light control. This show that active NR is not needed for nitrate uptake as suggested by Rao et al. (1976 ). Even in the presence of sucrose in the dark, or on exposure to white light for 5 min there was no difference in the nitrate content.
The nitrate reductase extracted from excised lintil leaves, incubated with 100 mM nitrate was passed through a Sephadex G-25 column to obtain the crude enzyme without nitrate concentration. Samples, whose nitrate content had been determined, were dried and the nitrate was extracted by boiling the samples in water. The solution was made up to the required volume and extract was then used as the substrate for NR, which was prepared by passing the solution through a Sephadex G -25 column, then incubating it for 2 h at 30°C, after which nitrate formed in the reaction was measured.
Decay of nitrate reductase in light and in the dark: The low level of NR induced in the dark may be due to the fast decay of the enzyme in the dark; there may be no difference in the rate of synthesis under either condition. The decay rate studied in light and in the dark showed that the half-life in the dark was more than the light, 16 and 18 h, respectively (Fig. 4). This means that enzyme synthesis in light is several times faster than in the dark. Similar results in the pea was faster in the dark (Sihag et al., 1979).
It is known that no nitrate reduction takes place in the dark, yet no accumulated nitrite is found in the dark in plants having a high NR level. This means that nitrate reduction also stop at the onset of darkness. Recently, Sawhney et al. (1978) studied nitrate reduction in wheat leaves and proposed that in the dark, the adenylate change goes down and mitochondrial respiration takes over, thus making NADH unavailable for nitrate reduction. Therefore, according to them, through the interaction of ATP, light and mitochondrial respiration, the process of nitrite reduction is controlled in plant systems.
According to Sluiters-Scholoten, (1975), the induction of NR by nitrate in bean leaves is influenced by the presence of reduction equivalents; also NADH may have a protective effect on the enzyme.
The experimental data obtained in our present study on the effects of light and photosynthates can be explained, if we assume that either the presence of NADH and or the process of nitrate reduction itself play an important role in the NR induction. Although the addition of sucrose increased the NR level in the dark, the light effect could not be due to the provision of photosynthates since even in the presence of light concentration of sucrose the activity never equaled that of the control; also the short duration of light treatments brought about an appreciable increase in the NR level. Since it has been proposed that the NADH produced in the glycolytic pathway may be used for nitrate reduction (Klepper et al., 1971); the observed enhancement by sucrose in the dark could be indirectly due to the increased provision of NADH caused by simulation of glycolytic enzymes. DNP and arsenate, uncouplers of oxidative phosphorylation, would also indirectly restrict NADH availability (Lehninger, 1978) for nitrate reduction and thus affect NR synthesis. Alternatively, nitrate uptake could also influence NR synthesis. Nitrate uptake, reduction and NR synthesis are believed to be interrelated (Rao et al., 1976; Shander et al., 1976; Tompkins et al., 1978). But in wheat leaves, the presence of nitrate in tissues treated with tungstate shows that at least initially, nitrate reduction does not govern the entry of nitrate. After some time , since there is no NR activity, i.e., no nitrate reduction, nitrate uptake would stop in dark treated leaves as well as in the presence of tungstate. In light the incoming nitrate may be going to the metabolic pool where it is utilized for enzyme induction. Hence, in light, the synthetic rate of the enzyme is high since there is continuous nitrate influx and reduction. The process of nitrate reduction and the existence of two pools of nitrate may not represent mutually exclusive mechanisms operating to control NR synthesis. Furthermore, there is the suggestion that the process of transfer from storage to the metabolic pool may be mediated by photochrome (Aslam et al.,1976). Control of NR by phytochrome occurs in wheat leaves; its mechanism is being studied in detail.