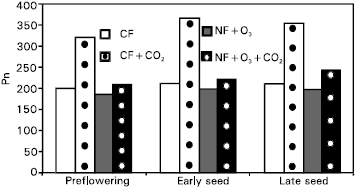
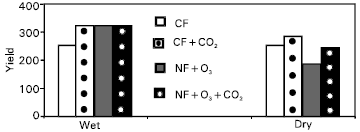
Research Article
Reversibility of Photosynthetic Inhibition After Long-term Exposure of Wheat Plants (Triticum aestivum L. Cvs. Sesquehanna and Gore) to Elevated Levels of Ozone
Department of Botany, Faculty of Science, University of Zagazig, Zagazig, Sharkia, Egypt
C. L. Mulchi
University of Maryland, Department of NRSLA, College Park, MD 20742, USA
I.A. Hassan
Department of Botany, Faculty of Science, University of Alexanderia, Alexanderia, Egypt
Y. A. El-Zawahry
Department of Botany, Faculty of Science, University of Zagazig, Zagazig, Sharkia, Egypt