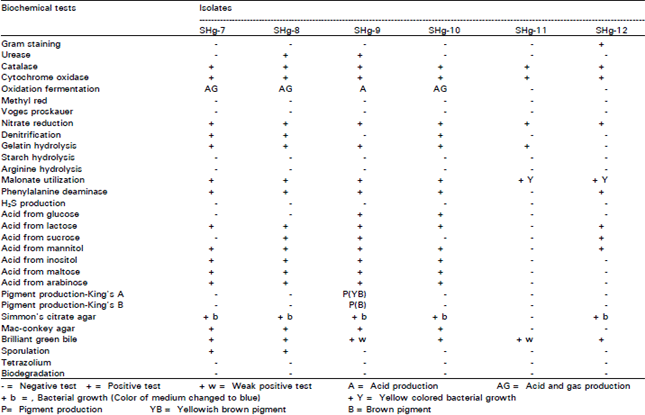
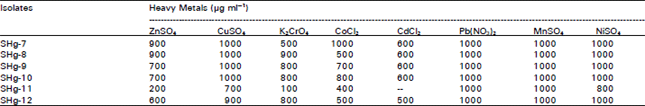
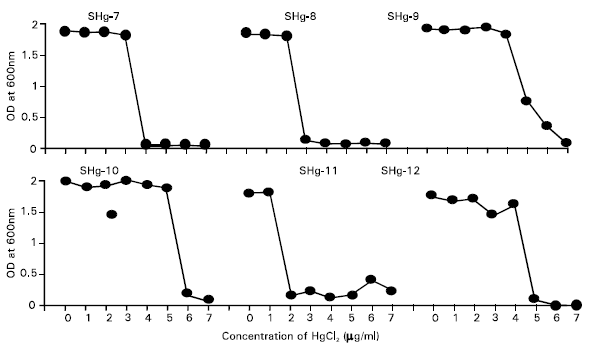
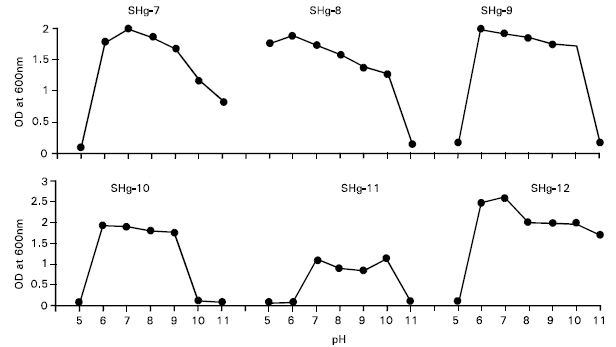
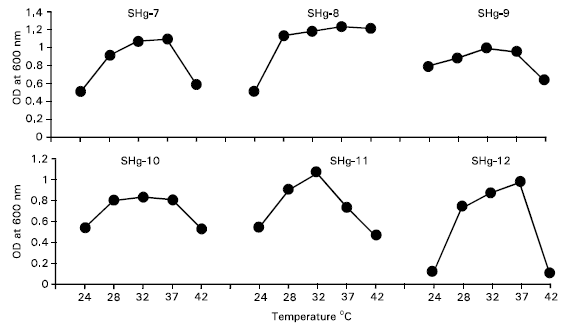
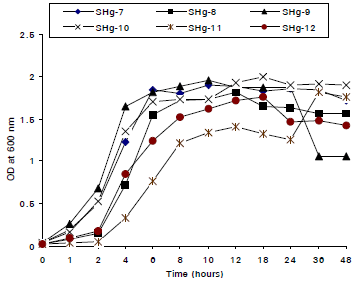
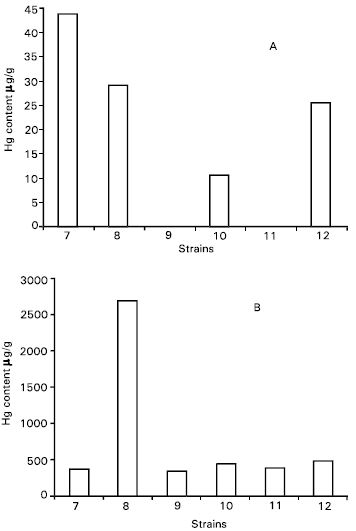
Research Article
Characterization of Some Indigenous Mercury Resistant Bacteria from Polluted Environment
Department of Botany, University of the Punjab, Quaid-e-Azam Campus, Lahore-54590, Pakistan
Azra Yasmin
Department of Botany, University of the Punjab, Quaid-e-Azam Campus, Lahore-54590, Pakistan
Shahida Hasnain
Department of Botany, University of the Punjab, Quaid-e-Azam Campus, Lahore-54590, Pakistan