Research Article
ALA-based Photodynamic Therapy of Jurkat Cells: Temperature Dependence
Department of Physics, Bangladesh Institute of Technology, Chittagong, Chittagong-4349, Bangladesh
Photodynamic therapy (PDT) is a novel form of treatment of cancer and other diseases based on the photosensitization of cells and their subsequent destruction on exposure to light (Kennedy et al., 1990 and Dahle et al., 2000). PPIX is an efficient photosensitizer participating in PDT reactions (Kennedy et al., 1990). The accumulation of PPIX in cells can be achieved by systemic administration of PPIX or systemic or topical δ-aminolevulinic acid (ALA) (Moan et al., 1999). The ALA is a precursor for the cellular biosynthesis of heme (Wyld et al., 1997 and Miah, 2001a). By adding exogenous ALA, PPIX is accumulated in cells and tissues. The administration of exogenous ALA bypasses the feedback effect of heme on the synthesis of ALA in the biochemical heme synthesis pathway and induces the accumulation of PPIX (Moan et al., 1999; Hilf et al., 1999; Jordan, 1990; Dailey, 1990 and Bogorad, 1979). Tumor cells appear to have a higher production of ALA-induced porphyrins than do normal tissues due to low activity of the ferrochetalase enzyme and probably also high activity of the rate-limiting enzyme porphobilinogen deaminase (PBGD) in the tumor cells (Hilf et al., 1999). Finally, ferrochetalase converts PPIX into heme by incorporating an iron atom into the porphyrin ring (Hilf et al., 1999; Dailey, 1990 and Bogorad, 1979). One apparent advantage for ALA-based PDT is the shorter duration of light treatment (Peng et al., 1997 and Kennedy et al., 1992). A critical consideration for effective treatment with ALA-based PDT is that the components of the heme biosynthetic pathway be fully functional and able to provide sufficient quantities of PPIX (Hilf et al., 1999; Kennedy et al., 1992 and Ades, 1990). Exposure to light results in a photochemical reaction that generates the reactive or toxic oxygen species, singlet oxygen, by energy transfer from excited triplet-state PPIX (Arakane et al., 1996). The ensuing oxidative-damage induced by the reaction of singlet oxygen with components in various subcellular locations leads to cytotoxicity and, ultimately, cellular damage or cell death (Melø et al., 1985). In this paper, human leukemic T cell line Jurkat (clone E6.1) has been studied under ALA-based PDT treatment at different incubation temperatures. A light source, using an array of light emitting diodes (LEDs) linked together, was constructed for this purpose.
This study was a one-year project and was carried out at the Department of Physics, Norwegian University of Science and Technology, Norway.
Chemicals: δ-Aminolevulinic acid hydrochloride (C5H9NO3.HCl) and trypan blue were purchased from Sigma Chemical Co. (Sigma-Aldrich, USA). A stock δ-Aminolevulinic acid (ALA) solution (100 mM) was prepared. The RPMI 1640 medium, L-glutamine, fetal calf serum (FCS) and gentamycine were from Merck, USA. A solution of ethanol (70%) was used to wash the Bürker Chamber.
Culture conditions: The Jurkat cell line (from the American Type Culture Collection, ATCC, USA) was cultured in an incubator (Biomed CO2-incubator, ASSAB Classic) at 37°C, 80% relative humidity and 5% CO2 conditions and maintained in a logarithmic phase. The medium RPMI 1640 was supplemented with fetal calf serum (FCS) (10%), L-glutamine (2mM) and gentamycine (40μg/ml). For keeping the cells in the logarithmic phase within the cellular concentration of 105-106 cells/ml, the cell suspension was diluted every 3rd or 4th day during the culture. In all experiments, the cells were treated under sterile conditions. The medium and flask were changed weekly to prevent the cells from poisoning in culture flask.
Irradiation system: Blue LEDs (Model: EL383UBC/H2) were purchased from the ELFA, Norway. An array of LEDs (14 LEDs x12 LEDs) linked together was constructed. The detailed description of construction of LED array, irradiation system and their coupling scheme is reported elsewhere (Miah, 2001b). The light system was calibrated with an IL 1700 light meter. A thermocouple thermometer (Digisense, Chicago, USA) was used to ensure that no detectable heating effect occurred during treatment of cells.
Irradiation: Cells incubated with 1 mM ALA for 4 hours were irradiated at room temperature (about 20°C) using the LED array irradiation system. The emission from LED was mainly in the wavelength region 400-500nm, with the highest fluence rate around 430nm. The light intensity reaching the cells was 2.5mW/cm2. After irradiation, cells were again incubated for 4 hours. The same procedure was followed in incubation at different temperatures. Cell survival was then studied by measuring the colony-forming ability of the cells.
Cell viability assay: A sample of 100 μl of the cell suspension (with cell density 1-2x106 cells/ml) was mixed properly with 50μl of 0.1% trypan blue (in 0.81% sodium chloride and 0.06% potassium phosphate solution). Trypan blue is excluded from viable cells, whereas non-viable cells with impaired membrane integrity take up the dye and appear blue on light microscopy (Jacob et al., 1985). Two drops of the resultant solution was placed on the plate of Bürker Counting Chamber, and the cells were counted by a microscope with required magnification.
The slope of the dose-response curve is higher for higher incubation temperature (Fig. 1). This means that the efficiency of cell inactivation is higher at higher temperatures, in general. The higher slope obtained at higher temperatures indicate that at this temperature range the rate or efficiency of cell inactivation is rapid.
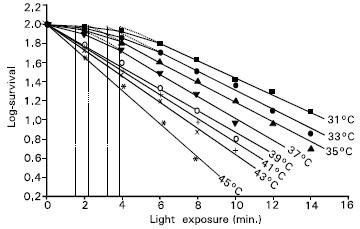 | |
| Fig. 1: | Temperature dependence of the log-surviving fractions of the Jurkat cells on light exposure. Each data represents the average over six measurements of independently performed experiments. A curve is fitted to the data for each incubation temperature. The threshold values calculated from the point of intersection of the straight survival curves at the survival in culture are 3.9, 3.2, 2.2 and 1.6min for temperatures 31, 33, 35 and 37°C, respectively. |
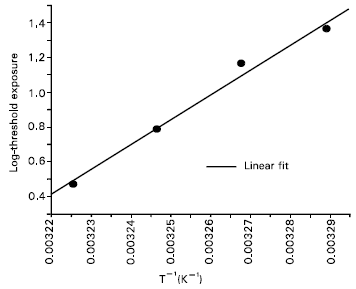 | |
| Fig. 2: | Arrhenius plot of the data for the temperature interval 31-37°C. The activation energy was calculated from the slope of the fitting (R: 0.99, P: 6.9 x 10-3) by using equation (1). |
This may be due to the contributions from heating effects for cell materials and cell surrounding's medium during incubation as well as from the accumulation of PPIX, as a photosensitizer, from ALA in the cell system (Moan et al., 1999; Gottfried et al., 1991 and Patrice et al., 1979).
The dose-response curves for lower incubation temperatures (31-37°C) have shoulders at lower exposure regions. For each temperature in the range from 31 to 37°C, a specific critical light dose or exposure time was detected. The phototoxic effect with this light exposure is small. This threshold value is defined as point of intersection of the straight survival curves with the survival in culture. The calculated threshold values were 3.9, 3.2, 2.2 and1.6min. for 31, 33, 35 and 37°C temperatures, respectively.
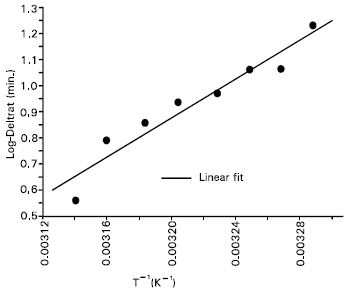 | |
| Fig. 3: | Plot of logeΔt versus 1/T for the temperature interval 31-45°C. A slope value of the light exposure (Δt) was calculated from each log-survival curve in Figure 1. The activation energy was calculated from the slope of the fitting (R: 0.96, P: 1.3 x 10-4) by using equation (1) (after replacing logetth by logeΔt). |
The data for threshold values can be analyzed in Arrhenius plot of the equation:
| (1) |
Discussion
The ALA-based phototoxic effect on Jurkat cells was studied for the first time. In the cell system, endogenous PPIX is produced from ALA that is taken up by the cells in suspension via cell wall receptors and stored intracellularly (Berg et al., 1990). Cytotoxicity induced by porphyrins in combination with light is due to photodynamic process. Porphyrin-induced photodestruction of cells results from reaction with singlet oxygen generated by energy transfer from excited triplet-state porphyrin in cell system (Dahle et al., 1999). Since porphyrins localize preferentially in membrane systems according to their lipophilicity (Peng et al., 1997), the plasma and organelle membranes are considered to be important targets in photodamage (Peng et al., 1997). Both proteins and lipids of the membrane can be damaged by porphyrin photoreactions. Photooxidation of these molecules results in an alternation of enzyme activities and transport functions.
Temperature dependent PDT treatment was to the cells. The result at lower incubation temperatures shows that the cells seems to have resistance against ALA-induced phototoxicity at these temperatures, indicating that some repair processes in the cells have been occurred. Shoulders in dose-response curves indicate that there should be some threshold exposure values for overcoming the resistance against phototoxicity. The cell inactivation was higher at higher incubation temperatures. The shoulders on the survival curves observed at temperatures 31-37°C was practically absent for incubation at higher temperatures. The observations may be related to a reduced repair of PDT-induced damage at higher temperatures. A pH dependent PDT treatment on human colon carcinoma cells has been carried out by Ma et al. (1999), where photosensitizing drug meso-tetrahydroxyphenyl chlorin (mTHPC) was used in their experiments. They have shown (Ma et al., 1999) that the shoulder on dose-response curves observed irradiation at 37°C and pH 7.2-7.8 completely disappeared for irradiation at lower temperatures (4°C) and even at 37°C when the light exposure was took place at lower pH, pH 6.8. Their results (Ma et al., 1999) say that under the same conditions the sensitivity of the colon carcinoma cells increases with decreasing pH value.
The production of ALA-induced PPIX in cells and tissues has been found to increase with temperature during light exposure (Moan et al., 1999 and Juzenas et al., 1999). Moan et al. (1999) have reported that the formation of PPIX in skin, both human and mouse, during typical application of ALA is temperature dependent, though the penetration of ALA into the skin is temperature independent. They (Juzenas et al., 1999) also declared that no PPIX was formed in the skin at lower temperatures (12°C). The activation energies calculated by them (Moan et al., 1999) for ALA uptake by cells, PPIX production in cells, ALA penetration into mouse skin, PPIX formation in mouse skin and PPIX production in human skin were 10, 13, 0.1, 17.2 and 17kcal/mol, respectively. In present work, activation energy of 25 kcal/mol was calculated for overcoming shoulder reactions in human leukemia cell type incubated with 1mM ALA for 4 hours. The activation energy of 9kcal/mol obtained for the cell system may be for ALA uptake by cells or PPIX production in cells, since this value is near the range as found by Moan et al. (1999) for these processes. This is, of course, slightly lower compared to the values as found by Moan et al. (1999) separately for different processes in cell and skin. Up to date, there is no published result on activation energy for Jurkat cells; therefore it is not possible to compare our result with literature values.
Temperature dependent ALA-induced PPIX production in cells have also been studied by Dietel et al. (1996). They (Dietel et al., 1996) reported that when cell temperature was raised to 35-40°C, the rate of porphyrin formation was increased for both PPIX and water-soluble porphyrins (e.g., UP, CP, etc) types at incubation period of 3 hours and for 6 hours incubation periods, the production of the water-soluble porphyrins was continued in a quasi-linear manner (i.e., slight acceleration) with respect to the incubation time. Gottfried and Kimel (1991) have studied the temperature effects on photosensitized reactions and have shown that in the temperature range 15-45°C a twofold increase or more in reaction rate has been observed on chemical acceptors. (Patrice et al., 1979) demonstrated that the temperature during HPD (hematoporphyrin derivative) treatment greatly influenced photocytotoxicity on both normal progenitors and leukemia cells. They (Patrice et al., 1979) reported that temperature variation induced changes in cell metabolism, explaining why at 4°C (incubation and irradiation) metabolism and thus HPD uptake were slower than at 37°C.
It has been established that the efficacy of photodestruction of the cells depends on the amount of photosensitizing porphyrins present in cell system (Hilf et al., 1999). Though the penetration of ALA into the cells for inactivation is practically not temperature dependent, the production of PPIX is strongly temperature dependent (Moan et al., 1999 and Juzenas et al., 1999). Because PPIX is a photosensitizer, the efficacy of ALA for use in PDT depends on the accumulation of sufficient levels of PPIX in cells (Hilf et al., 1999 and Miah, 2002). Cool temperatures are likely to result in a higher pH (less cell metabolism) and decreased drug uptake. Both of these effects will decrease cell killing. For a better understanding this higher-ordered cell inactivation at higher temperatures, the effect of temperature alone should be demonstrated, pH in the medium measured and the amount of drug uptake in cells at various temperatures should be assessed.
The efficiency for cell inactivation was found to increase with increasing incubation temperature. The higher production rate of photosensitizing PPIX, more cell metabolism and increased drug uptake at higher temperatures might play an important role in the observed higher cytotoxic effect at higher incubation temperatures. The log-survival curves for incubation temperatures 31-37°C showed shoulders at lower exposure regions. The threshold values for the temperatures were calculated from the point of intersection of the straight survival curves at the survival in culture and was found to be 3.9, 3.2, 2.2 and 1.6min for 31, 33, 35 and 37°C, incubation temperatures respectively. The activation energies for overcoming the PDT-induced damage for the cell system and ALA uptake by cells (or PPIX production in cells) were 25 and 9kcal/mol, respectively.
This study was supported by the Norwegian Research Council for Science and Humanities through a fellowship to author. The author thanks to Professor A. Johnsson for his useful discussion.