Research Article
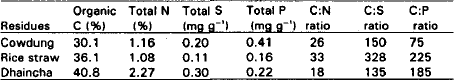
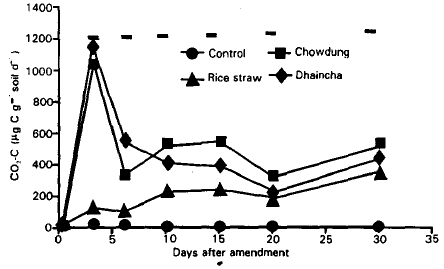
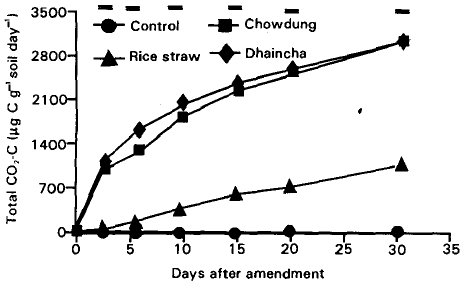
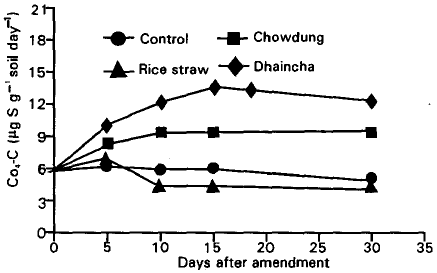
Plant and Animal Residue Decomposition and Transformation of S and P in Soil
Department of Agricultural Chemistry, Bangladesh Agricultural University, Mymensingh, 2202, Bangladesh
R. Begum
Department of Agricultural Chemistry, Bangladesh Agricultural University, Mymensingh, 2202, Bangladesh
M. R. Kabir
Department of Agricultural Chemistry, Bangladesh Agricultural University, Mymensingh, 2202, Bangladesh
H. M. Zakir
Department of Agricultural Chemistry, Bangladesh Agricultural University, Mymensingh, 2202, Bangladesh