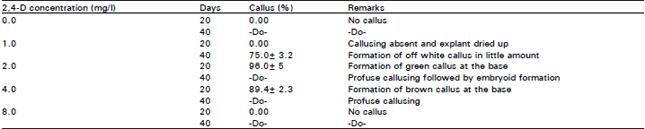
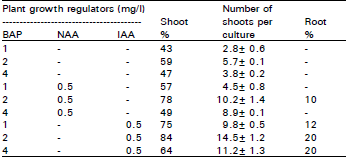
Research Article
Effects of Various Growth Regulators on Callus Formation and Regeneration in Brassica napus Cv. Oscar
Agricultural Biotechnology Institute, National Agricultural Research Center, Park Road, Islamabad, Pakistan
Hamid Rashid
Agricultural Biotechnology Institute, National Agricultural Research Center, Park Road, Islamabad, Pakistan
Azra Quraishi
Agricultural Biotechnology Institute, National Agricultural Research Center, Park Road, Islamabad, Pakistan