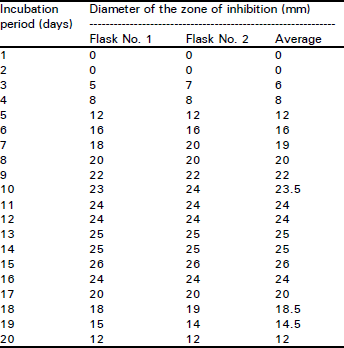
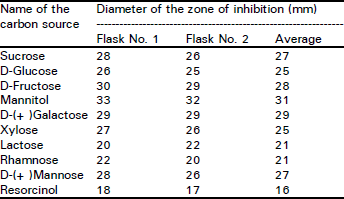
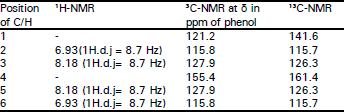
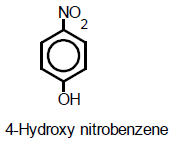
Research Article
Isolation of a Yellowish Antibiotic Pigment 4-hydroxy Nitrobenzene from a Strain of Streptomyces
Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
Naoki Sugimoto
National Institute of Health Sciences, Tokyo, Japan
Md. Ibrahim Khalil
Department of Biochemistry, University of Rajshahi, Rajshahi-6205, Bangladesh
M. A. Gafur
Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh