ABSTRACT
The present study evaluates the effects of environmental factors and associated species on the distribution and abundance of Zygophyllum aegyptium A. Hosny in different habitats in the Deltaic Mediterranean coast of Egypt. Four vegetation groups associated with Z. aegyptium were identified using Two way Indicator Species Analysis. These groups were named as Elymus farctus (Viv.) Runemark (group A), Alhagi graecorum Boiss. (group B), Silene succulenta Forssk. (group C) and Stipagrostis lanata (Forssk.) De Winter (group D).
The application of Detrended Correspondence Analysis and Canonical Correspondence Analysis explored the pattern of distribution and the main environmental factors influencing the distribution and abundance of Z. aegyptium. This showed the highest scores of abundance in the salt marshes (group A) and fertile non-cultivated lands (group B) however, the lowest scores of abundance in the sand dunes (groups C&D). Edaphically, the distribution of Z. aegyptium is mainly controlled by soil texture, moisture availability, calcareous sedimentation and soil fertility (organic carbon).
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2002.152.160
URL: https://scialert.net/abstract/?doi=pjbs.2002.152.160
INTRODUCTION
The landscape of the Deltaic Mediterranean coastal land of Egypt is differentiated into: sand formations, salt marshes, fertile non-cultivated lands and reed swamps. The sand formations are mainly composed of silecous deposits and they are distinguished into three subhabitat types: sand mounds, sand sheets and sand dunes. The sand mounds occupy the frontal belt of the zonation pattern in the Deltaic coast; they are either low, medium or large sized. The sand sheets are irregularly scattered between sand dunes and salt marshes and they are either saline or non-saline sand flats. The sand dunes are the main geomorphic features in the greatest part of the Deltaic coast. These dunes are classified into three kinds: mobile sand dunes, partial stabilized sand dunes and stabilized sand dunes which are usually of varying sizes and heights. The salt marshes are characterized by a shallow water table and/or high level of salinity. Some of these occur around edges of Lake Manzala, Lake Burullus and Lake Idku and their dried bed and others are found close to the Mediterranean Sea and are thus periodically inundated by sea water. Also, there are salt marshes in the depressed areas between sand dunes. These are usually wetted by water seeped from the sea and lakes especially during winter season. The fertile non-cultivated lands are found on the well drained less saline soils. They are formed by transportation of sands from the shifting sand dunes to neighbouring low lands. Some patches of this habitat type have been farmed with many vegetables, crops and orchards. The reed swamps are frequent in the Deltaic Mediterranean coast. They are formed by accumulation of water seeped from the northern lakes, Mediterranean Sea and/or drainage systems of the Delta in depressed areas.
Vegetation analysis and ecological studies in the Deltaic Mediterranean coast reveal that many species are of wide ecological amplitude; each attains a relatively high value of abundance in at least two habitat types (Zahran et al., 1990; El-Demerdash et al., 1990; Shaltout et al., 1995; Mashaly 2001). Zygophyllum aegyptium is one of the most important taxon of these species in this region. It is a perennial woody undershrub with evergreen succulent leaves. The species is distributed in the Mediterranean region of Egypt, Tunis and Cyprus. The species is described by Hosny (1977) and Hadidi (1978) and it has a broad ecological amplitude.
Hitherto, a few reports are available on the ecology of Z. aegyptium. Previous studies focused on the taxonomic description (Hosny, 1977; Hadidi, 1978), cytological studies (Soliman, 1995; Ahmed, 2001) and the range of environmental requirements for seed germination (Mashaly et al., 1993). Therefore, the main objective of the present study is to analyze the distributional behaviour of Z. aegyptium in order to: provide quantitative assessment of its abundance in different habitats in the Deltaic Mediterranean coast and to estimate the magnitude of variation of such abundance within each habitat, evaluate the correlations with different edaphic factors and to determine the mode of its distributional behaviour along the gradients of controlling environmental factors.
MATERIALS AND METHODS
The study area: The study area is the Deltaic coastal sector of the Mediterranean region of Egypt. It forms a belt extending in the E-W direction for about 180 Km between Port-Said to Abu-Qir, and from the N-S direction for an average of 10 Km from the coast, i.e. the study site covers an area of about 1800 Km (Fig. 1).
According to the analysis of the Neogene-Quaternary section in the Nile Delta, Rizzini et al. (1979) proposed a model of elastic sedimentation in the Delta area, which consists of three sedimentary cycles: Miocene, Plio-Pleistocene and Holocene. The Deltaic coastal section is basically formed of different particle sizes of sand, silt and clay deposited by the River Nile (Abu Al-Izz, 1971). It is characterized by three shallow brackish natural northern lakes: Manzala, Burullus and Idku. The study area is included in Emberger's Mediterranean isoclimatic zone (Houerou, 1981) and the warm coastal deserts (Meig, 1973). The bioclimatic map of UNESCO (1979) designates its climate as arid with a mild winter and a warm summer.
Sixty stands (13 x 13 m2) representing different habitat types of Z. aegyptium were selected along the Deltaic Mediterranean coastal lands of Egypt. These stands were chosen to cover apparent variations in the physiognomy of vegetation and in the physiographic features. Disturbances caused by particular human impacts were avoided as far as possible. The sampling process was carried out during April-November 2000, the flourishing period of vascular plant richness and species diversity. In each stand, relative density and relative frequency were estimated quantitatively using the point-centered quarter method (Cottam & Curtis, 1956). While, relative cover was estimated by applying the line intercept method (Canfield, 1941). Species abundance as expressed by relative values of density, frequency and cover were calculated for each perennial species and summed up to give an estimate of its importance value. The associated annual species in each stand were only recorded. Plant specimens were collected, identified and preserved at the Herbarium of Botany Department, Faculty of Science, Mansoura University. The life-forms were according to Raunkiaer (1934). Species identification and floristic categories were according to Meikle (1977 & 1985); Feinbrun-Dothan (1978 & 1986) and Boulos (1995, 1999 & 2000).
Four soil samples (0-50 cm) were collected from each stand, pooled together to form composite sample, air dried, passed through 2 mm sieve to remove gravel and debris, and packed in plastic bags ready for physical and chemical analysis. The procedures followed in estimating the soil variables were according to Piper (1947); Jackson (1962) and Allen et al. (1974).
Vegetation was classified using TWINSPAN (Two-Way Indicator Species Analysis) and ordinated using DCA (Detrended Correspondence Analysis, Hill, 1979a&b). The abundance scores (importance values) of Z. aegyptium and associated species in the studied stands in relation to the environmental factors were analyzed using Canonical Correspondence Analysis (CCA). The result is an ordination diagram (CCA biplot), on which points represent species and arrows represent environmental variables (Ter Braak, 1988). Statistical treatments applied in present study were according to Snedecor & Cochran (1968) and Anonymous (1993).
RESULTS
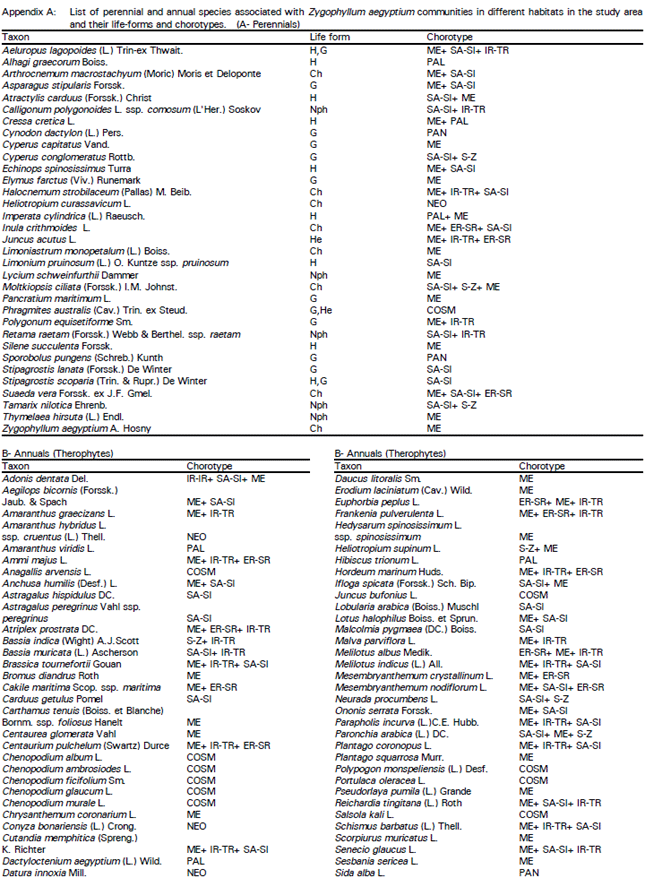
The present investigation reveals that, the natural plant wealth of the study area is composed of 109 species divided into 33 perennials and 76 annuals (Appendix). These recorded species are classified into five main types of life-forms: therophytes (69.7%), cryptophytes comprising geophytes and helophytes (10.1%), hemicryptophytes (8.3%), chamaephytes (7.3%) and nanophanerophytes (4.6%). The floristic analysis of the recorded species indicates that they are mainly of Mediterranean origin (67 species or about 61.5%) and partly of Cosmopolitan element (14 species or 12.8%) and of Tropical origin (11 species or 10.1%). A further 17 species (15.6%) are belonging to different chorotypes with poor representation.
In the study area, the most common perennial species in Z. aegyptium communities are: Alhagi graecorum, Stipagrostis lanata, Arthrocnemum macrostachyum and Echinops spinosissimus; each recorded in 16 or more stands (Table 1).
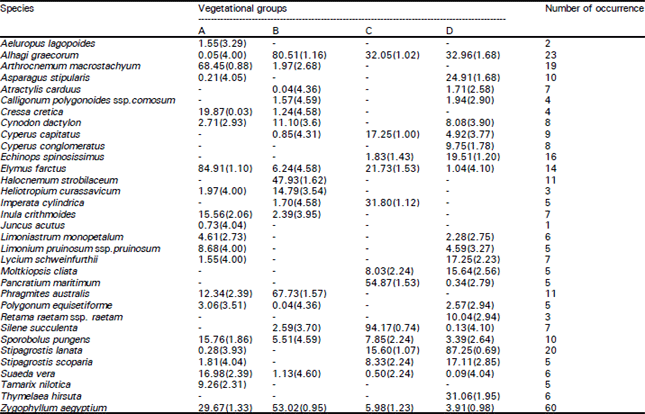
| Table 1: | Mean and coefficient of variation (values between brackets) of the importance values (out of 300) of Zygophyllum aegyptium and the associated perennial species in different vegetation groups resulting from classification technique of the sampled stands in the Deltaic Mediterranean coastal lands of Egypt. |
 | |
| Table 2: | Mean value and standard error of the soil variables in different TWINSPAN vegetation groups of Zygophyllum aegyptium in the Deltaic Mediterranean coastal land of Egypt. |
 | |
| 10.85±1.88WHC = Water-holding capacity, EC = Electrical conductivity, SAR = Sodium adsorption ratio, PAR = Potassium and adsorption ratio. | |
| Table 3: | Pearson-moment correlation (r) between different soil variables in the stands of Zygophyllum aegyptium surveyed in the study area. |
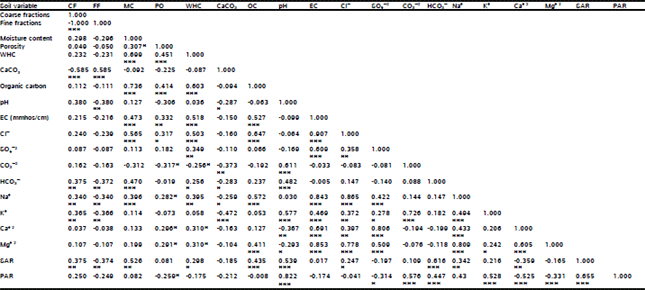 | |
| Abbreviation: CF = Coarse fraction, FF = Fine fraction, MC = Moisture content, Po = Porosity, WHC = Water holding capacity, OC = Organic carbon, EC = Electrical conductivity, SAR = Sodium adsorption ratio, PAR = Potassium adsorption ratio. * is significant at P ≤ 0.05, ** is significant at P ≤ 0.01, *** is significant at P ≤ 0.001. | |
The classification of stands according to TWINSPAN technique resulted in four vegetation groups (Fig. 2) and the vegetation structure of these groups is presented in Table 1, and the soil variables are presented in Table 2.
Group A: Sixteen stands of the salt marsh and saline sand flat habitats were dominated by the preferential halophyte grass Elymus farctus, which attains the highest mean importance value (IV) of 84.91 (out of 300). Indicator species were the obligatory halophytes: Arthrocnemum macrostachyum (IV = 68.45) and Suaeda vera (IV = 16.98). Other important species attaining relatively high IV were: Z. aegyptium (IV = 29.67) and Cressa cretica (IV = 19.87). The soil of this group (A) was characterized by relatively higher values of moisture content, organic carbon, electrical conductivity, chlorides, sulphates, bicarbonates, extractable cations (Na+, K+, Ca+2 & Mg+2) and sodium adsorption ratio.
Group B: Twenty one stands of the fertile non-cultivated lands were dominated by salt-tolerant indicator species Alhagi graecorum (IV = 80.51). In this group, the second indicator species was Phragmites australis (IV = 67.37). The important species were also Zygophyllum aegyptium (IV = 53.02) and Halocnemum strobilaceum (IV = 47.93). The soil was characterized by high percentages of fine fractions (silt & clay) and calcium carbonate content, moderate values of EC, sulphates and calcium cation; and low contents of organic carbon, extractable cations (Na+, K+ & Mg+2) as well as SAR and PAR.
Group C: Six stands of the partially stabilized sand dunes were dominated by psammophytic succulent indicator species Silene succulenta (IV = 94.17). The second indicator species in this group was the grass Imperata cylindrica (31.80), and the other important species were Pancratium maritimum, Alhagi graecorum and Elymus farctus, which attain IV of 54.87, 32.05 and 21.73 respectively.
 | |
| Fig. 1: | Map showing the Deltaic (middle) section of the Mediterranean costal region of Egypt. *Hatched area indicates the location of the study area. |
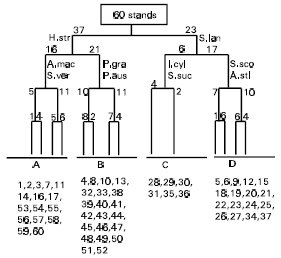 | |
| Fig. 2: | TWINSPAN dendrogram of the 60 stands based on the importance values of 33 species representing the different habitats of Z. aegyptium. Indicator species are abbreviated to the first letter of the genus and the first three letters of species names respectively. For full names of species see Table 1. |
The soils of this group were obviously characterized by low contents of most soil variables such as moisture content, calcium carbonate, EC, sulphates and calcium cation. While, these soils attained moderate values of Na+ & K+ as well as SAR and PAR.
Group D: Seventeen stands of the stabilized sand dunes were dominated by psammophytic grass Stipagrostis lanata (IV = 87.25). Here the indicator species were Asparagus stipularis (IV = 24.91) and Stipagrostis scoparia (IV = 17.11). Other important species which attain relatively high IV in this group were Alhagi graecorum (IV = 32.96), Thymelaea hirsuta (IV = 31.06) and Echinops spinosissimus (IV = 19.51). The soils of group D were markedly characterized by relatively higher contents of most edaphic factors such as coarse fractions (sand), organic carbon, EC, chlorides, sulphates and extractable cations (Na+, K+, Ca+2 & Mg+2).
It is interesting to note that, Z. aegyptium showed the highest scores of abundance in vegetation groups A and B which represent the habitats of salt marshes and fertile non-cultivated lands. On the other hand, it showed the lowest scores in groups C and D where these two groups represent the habitats of sand dunes in the study area.
It is also worth to recognize that, the soils of groups A and D seem to be more or less comparable, whereas the soils of groups B and C also showed a close similarity between each other. The two later groups of soils (B & C) may be considered as transitional state between the two former groups of soils (A & D).
In the present study, the most common associated annuals comprise: Aegilops bicornis, Anchusa humilis, Bassia indica, Bromus diandra, Cakile maritima ssp. maritima, Carthamus tenuis ssp. foliosus, Chenopodium glaucum, Cutandia memphitica, Daucus litoralis, Heliotropium supinum, Lobularia arabica, Malcolmia pygmaea, Parapholis incurva, Paronchia arabica, Pseudorlaya pumila, Sphenopus divaricatus, Suaeda maritima, Trisetaria linearis, etc. (Appendix).
The correlation coefficient (r) between different environmental variables in the sampled stands supporting the growth of Z. aegyptium are shown in Table 3. All soil variables showed positive and negative significant correlations between each other except for moisture content, porosity and pH which showed a limited significant correlations with the other edaphic factors.
Detrended Correspondence Analysis (DCA) produced a marked distinguishable separation of the salt marshes and saline sand flats (stands of group A), from those of the fertile non-cultivated lands (stands of group B), and from the partial stabilized sand dunes represented by stands of group C, as well as from the stabilized sand dunes represented by stands of group D (Fig. 3a). The DCA-ordination of the species as shown in Fig. 3b exhibited a clear pattern of segregation on the first and second ordination axes. On DCA-ordination diagram, Z. aegyptium is distributed between the sites of vegetational groups A and B, this distribution may be prove that, this plant species prefers to germinate and grow in the habitats of the dry salt marshes, saline sand flats and fertile non-cultivated lands rather than in the habitats of the sand dunes.
The ordination diagram produced by Canonical Correspondence Analysis (CCA) is shown in Fig. 4. The position of Z. aegyptium and associated species along the gradient of 14 environmental variables was clear. The CCA-biplot diagram showed that calcium carbonate and fine fractions (silt & clay) of the soil were the most important factors related to the distribution of this species in the study sites. The distribution of Z. aegyptium was concentrated at the end of the lower gradient of the extractable cations (Mg+2, Ca+2, K+ & Na+), sulphates, chlorides, carbonates & bicarbonates and on the intermediate gradient of soil moisture content, SAR and organic carbon. The salt-tolerant species like Phragmites australis, Halocnemum strobilaceum, Calligonum polygonoides ssp. comosum and Alhagi graecorum showed similar trend of distribution to that of Zygophyllum aegyptium. However, a second group of associated species such as Tamarix nilotica, Polygonum equisetiforme, Cressa cretica and Aeluropus lagopoides showed an opposite trend of distribution to that of Zygophyllum aegyptium. The halophytic species: Suaeda vera, Arthrocnemum macrostachyum, Inula crithmoides, Elymus farctus and Juncus acutus were associated with soils of high moisture content.
DISCUSSION
The classification and ordination of vegetational units has in recent times received serious attention, and a number of theoretical and practical treatises have been published on this subject.
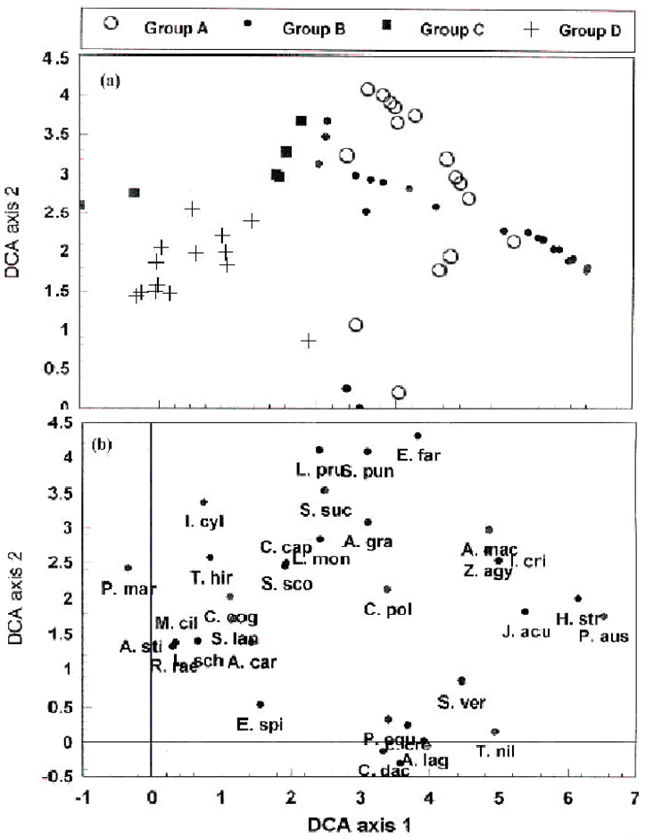 | |
| Fig. 3: | (a) DCA-ordination diagram of 60 stands of Z. aegyptium with TWINSPAN group superimposed. (b) DCA-ordination diagram of the species with the names abbreviated into the first letter of the genus and first three letters of the species. |
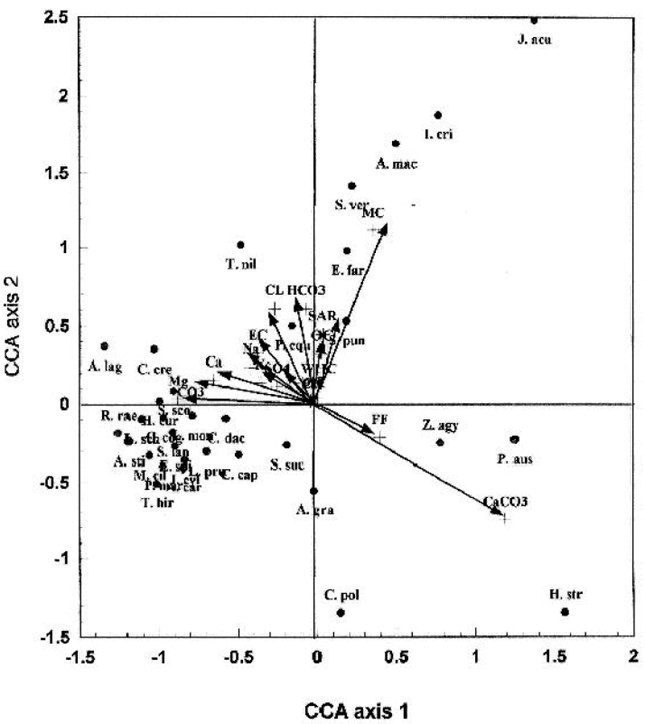 | |
| Fig. 4: | CCA-biplot of the indicator and preferential species associated with Z. aegyptium represented by the first letter of the genus and first three letters of the species (points) and edaphic variables (arrows). For complete names of the species see Table 1 or Appendix. |
The vegetation dealt in present study is classified by TWINSPAN classification into four groups named after their dominant species as follows: Elymus farctus (group A), Alhagi graecorum (group B), Silene succulenta (group C) and Stipagrostis lanata (group D). Most of the species of these groups have analogues in the western Mediterranean coastal region of Egypt (Tadros & Atta, 1958; Ayyad & Hilmy, 1974; Ayyad 1976; Ayyad & El-Ghonemy, 1976; Ayyad & El-Ghareeb, 1982) and in the middle Deltaic Mediterranean coastal land (Zahran et al., 1990; El-Demerdash et al., 1990; Shaltout et al., 1995 and Mashaly 2001).
 |
 |
The vegetational groups resulted from the application of multivariate analysis in the present work may be related to the alliances and associations described by Zohary (1973). Out of the identified clusters in present study, group A dominated by Elymus farctus may represents the dry salt marshes and saline sand flats in the study area. While, group B dominated by Alhagi graecorum may represents the fertile non cultivated lands. On the other hand, group C dominated by Silene succulenta represents the partial stabilized sand dunes, while group D dominated by Stipagrostis lanata represents the stabilized sand dunes. The soil analysis and vegetation structure of the sampled stands showed that groups B and C may be considered as a transitional habitats between the habitats of groups A and D. These transitional groups (B & C) may be formed by transportation of sand grains from the shifting huge sand dunes (group D) to neighbouring low lands of the salt marshes (group A) in the study area.
The spatial relations of species are evidently a product of their different needs and tolerances for physical and/or biotic factors (Ayyad & Hilmy, 1974). Many studies in different habitats in the western Mediterranean coastal lands of Egypt assert that few composite gradients are responsible for quantitative variations in the distribution of populations of species (Shaltout, 1983; Ayyad & El-Ghareeb, 1982; Ghareeb 1990).
The application of TWINSPAN classification showed that, the taxon Z. aegyptium has been estimated as associated species and not as dominant in the four recognized vegetation groups. This plant species showed the highest scores of abundance in groups A and B while, it showed the lowest scores of abundance in groups C and D. However, the application of Detrended Correspondence Analysis (DCA) and Canonical Correspondence Analysis (CCA-biplot) showed that, the distributional behaviour of Z. aegyptium is mainly related to the contents of calcium carbonate and fine fractions and partly to the soil moisture content and organic carbon content. This may illustrate the fact that, Z. aegyptium predominates and has a luxuriant growth with the intermediate gradient of the environmental factors prevailing in the intermediate habitats derived between the salt marshes and sand formations in the study area.
Therefore, it can be concluded that micro variations in the distribution behaviour of Z. aegyptium in the Deltaic Mediterranean coastal land of Egypt is controlled edaphically by four main factors: soil texture (fine fractions), moisture availability (moisture content), calcareous sedimentation (calcium carbonate) and soil fertility (organic carbon).
REFERENCES
- Allen, S.E., 1974. Chemical Analysis of Ecological Materials. Blackwell Scientific Publications, London, UK., Pages: 565.
Direct Link - Ayyad, M.A. and R.E.M. El-Ghareeh, 1982. Salt marsh vegetation of the Western Mediterranean Desert of Egypt. Vegetatio, 49: 3-19.
CrossRefDirect Link - Canfield, R.H., 1941. Application of the line interception method in sampling range vegetation. J. For., 39: 388-394.
Direct Link - Cottam, G. and J.T. Curtis, 1956. The use of distance measures in phytosociological sampling. Ecology, 37: 451-460.
CrossRefDirect Link - El-Demerdash, M.A., M.A. Zahran and M.S. Serag, 1990. On the ecology of the deltaic Mediterranean coastal land, Egypt. III. The habitat of salt marshes of Damietta-Port Said coastal region. Arab Gulf J. Scient. Res., 8: 103-119.
Direct Link - Mashaly, I.A., 2001. Contribution to the ecology of the deltaic mediterranean coast, Egypt. J. Biol. Sci., 1: 628-635.
CrossRefDirect Link - Mashaly, I.A., A.K. Hegazy and M.I. Soliman, 1993. On the Ecology of Zygophyllum aegyptium in Egypt. In: Towards the Rational Use of High Salinity Tolerant Plants, Volume 1: Deliberations about High Salinity Tolerant Plants and Ecosystems, Lieth, H. and A.A. Masoom (Eds.). Kluwer Academic Publishers, UK., ISBN: 9780792318651, pp: 423-433.
- Piper, C.S., 1947. Soil and Plant Analysis. 1st Ed. Interscience Publishers Inc., New York, USA.
Direct Link - Raunkiaer, C., 1934. The Life Forms of Plants and Statistical Plant Geography. Clarendon Press Oxford, London, United Kingdom, Pages: 632.
Direct Link - Shaltout, K.H., H.F. El-Kady and Y.M. Al-Sodany, 1995. Vegetation analysis of the Mediterranean region of Nile Delta. Vegetation, 116: 73-83.
CrossRefDirect Link - Tadros, T.M. and B.A.M. Atta, 1958. The plant communities of barley fields and uncultivated desert areas of Mareotis (Egypt). Vegetatio, 8: 161-175.
CrossRefDirect Link - Ter Braak, C.J.F., 1988. CANOCO-A FORTRAN program for canonical community ordination by (partial) (detrended) (canonical) correspondence analysis, principal component analysis and redundancy analysis (Version 2.1). Ministry of Agricultural Mathematics Group, Technical Report LWA-88-02, Wageningen, Netherlands.
- Zahran, M.A., M.A. El-Demerdash and I.A. Mashaly, 1990. Vegetation types of the deltaic Mediterranean coast of Egypt and their environment. J. Vegetation Sci., 1: 305-310.
CrossRefDirect Link








