ABSTRACT
The objective of this study was to determine the interrelationships among various factors governing physiological responses of soybean [Glycine max (L.) Merr.] leaves to different water table (WT) depths (0.15, 0.30, 0.45 and 0.60 m below the surface). Photosynthesis, stomatal conductance, transpiration, chlorophyll and leaf temperature were measured weekly on leaflets of soybean (cv. Hobbit 87) grown in lysimeters in controlled environment chambers. A week after the WT treatments were established, the photosynthesis and transpiration rates decreased significantly with the rise in WT depth. However, the results were not found consistent during the WT treatment period of this study. Mean photosynthetic rates and stomatal conductance for 0.15 m WT treatment were 19 and 10% lower than 0.60 m WT treatment. Effects of WT on chlorophyll content was not noticed until later part of the growing season, where significantly lower values of this parameter were observed for 0.15 m WT treatment than 0.60 m WT treatment. Leaf transpiration was highly responsive to WT depth, showing significant differences between WT treatments. Canopies under 0.15 m WT treatment transpired 16% less water compared with 0.45 m WT treatment due to excessive moisture stress. Shallow water table depths significantly reduced the grain yield compared with deeper WT depths. Highest soybean yield was obtained under 0.60 m WT depth and the lowest was for 0.15 m WT depth. Average yield obtained at 0.15 m WT was 48% lower than 0.60 m WT treatment. Relationships between different parameters were developed by various regression models. Linear positive relations were found between photosynthesis and leaf temperature under deeper WT depths. Results of these analysis revealed that leaf temperature was a good parameter to predict photosynthetic rates. The transpiration rate was found to be highly dependent on stomatal behaviour. Seed yield was also increased linearly with the increase in photosynthesis, stomatal conductance and transpiration rate. Overall results of this study show that plant physiological parameters could be used to evaluate the effectiveness of WT management practices for soybean growth.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2002.1300.1308
URL: https://scialert.net/abstract/?doi=pjbs.2002.1300.1308
INTRODUCTION
Crop production is based on photosynthesis. Improvement in the photosynthetic mechanism has become one of the major research interests of recent decades. Many environmental factors, such as photosynthetic active radiation (PAR), CO2 concentration and water status of plants and soil, affect photosynthesis as reported by many researchers. It has been shown that water stress reduced the rate of CO2 assimilation and leaf conductance in corn during a period of 14 days when leaf water potential decreased from 0.5 to 8.0 bars (Wong et al., 1985). Huber et al. (1984) reported that water stress reduced carbon exchange rate (CER) of soybean and the reduction was greater in non-CO2 enriched plants than in CO2 enriched plants. As CER declined, stomatal resistance increased, but this was not the primary cause of decrease in assimilation because CO2 concentration remained relatively constant.
Many researchers have been interested in understanding how water stress limits the photosynthesis. Mohanty and Boyer (1976) and Mooney et al. (1977) reported that quantum yield of photosynthesis was decreased at low water potential. Photo-respiratory CO2 evolution was also shown to decrease at low water potential in wheat and sunflower (Lawlor, 1976; Mohanty and Boyer, 1976). In addition, photosynthetic electron transport chain activity was inhibited in plants subjected to water stress (Boyer and Bowen, 1970; Keck and Boyer, 1974). Nicolodi et al. (1988) determined photosynthesis of the upper leaves of irrigated and non-irrigated alfalfa (cv. Dabina) for two years. In both years, net photosynthesis (Pn) was inhibited more than 35% in non-irrigated plants compared to irrigated plants, which had higher leaf water potential. Hesketh (1963) showed that plant species can vary greatly in rate of photosynthesis, but this variation is not related to variation of chlorophyll content. Kariya and Tsunoda (1972) also reported that the rate of photosynthesis was not necessarily related to chlorophyll contents, at least under high light.
Some investigators have conducted studies on soybean root and yield response to WT control (Reicosky et al., 1972; Cooper et al., 1992), but limited research has been conducted to understand the physiological response (like photosynthesis, stomatal conductance, chlorophyll content etc.,) of soybean to shallow water tables. Therefore, the objective of this study was to determine the effects of shallow water tables on physiological parameters of soybean in controlled environmental conditions.
MATERIALS AND METHODS
The study was conducted in two controlled environment growth chambers at the Iowa State University, Ames, U.S.A during 1995. Water tables (WT) were maintained at 0.15, 0.30, 0.45 and 0.60 m below the soil surface and were replicated four times. In order to control the water table 16 portable lysimeters were constructed (Fig. 1). The lysimeters were made from plastic containers (52x42x71 cm3). Detailed procedures of the construction of lysimeters are given by Sarwar and Kanwar (1996). These lysimeters were placed in the controlled environment growth chambers. Each growth chamber could hold 8 lysimeters. Daily diurnal temperatures inside the growth chambers were programmed to simulate Ames climatic conditions between May 01 and September 30. These temperature values were based on 30-years (1965-1994) maximum and minimum temperatures for the corresponding dates. The temperatures were ramped between hourly set points. For the variable daylight periods, light was provided by 45 incandescent 120-W and 30 fluorescent 115-W light bulbs. During the first hour, only incandescent light was used and for the last hour, only fluorescent light was provided. Relative humidity in these chambers could not be controlled, however, its values were above 50% for most of the growing season.
Experimental design and layout: The experimental treatments consisted of WT levels maintained at 0.15, 0.30, 0.45 and 0.60 m below the soil surface (these treatments started on 37 DAP and ended on 107 DAP). These treatments were arranged in a randomized complete block design each with four replications. There were two replicates in each growth chamber. Lysimeters were arranged in the growth chambers in such a way that each lysimeter received similar light level.
Data on photosynthetic rate, stomatal conductance, transpiration rate, chlorophyll content and leaf temperature were collected weekly. Four middle plants in each lysimeter were used for measurements. Thus, the mean values obtained for each of these parameters were the average of 16 plants.
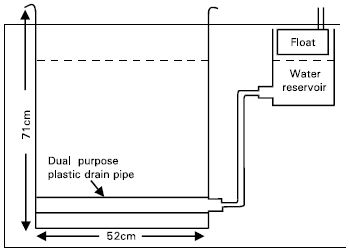 | |
| Fig. 1: | Schematic sketch of a lysimeter |
The chlorophyll content was also determined weekly by chlorophyll meter. The same four plants and leaves were used for chlorophyll determination. The latest fully developed leaf (generally 3rd or 4th leaf from top) was used for chlorophyll measurements. Six measurements were made on each plant using the same leaf. Thus, the mean values obtained for chlorophyll content was the average of 384 measurements.
Planting: In the center of each lysimeter, a groove was made about 20 cm from the walls of lysimeter. Eighteen soybean (cv. Hobbit 87) seeds were planted in each lysimeter. After germination these plants were thinned to six plants per lysimeter. The same procedure was repeated in both growth chambers. Before planting, soil samples were taken from all lysimeters to determine the soil fertilizer needs. The results of soil analysis showed high amounts of phosphorus and potassium already present in the soil; therefore, no fertilizers were applied.
Irrigation: To determine the amount of water required for each surface irrigation during the growing season, thirty years (1965-1994) of rainfall data were obtained and weekly averages were calculated from May 01 through September 30. To simulate field conditions, rainfall water was applied on a weekly basis (equal to 30 years average weekly rainfall) in the form of irrigation because rainfall intensities in the controlled environment growth chambers could not be obtained. This kind of irrigation schedule was needed to match climatic conditions for soybeans grown in the humid Midwest of the U.S.A.
Measurement of photosynthesis and other parameters: Measurements on photosynthesis, stomatal conductance, transpiration rate, leaf temperature and air temperature were made with a portable photosynthesis system (LICOR-6200). This system consists of a CO2 analyzer, a system console and a sensor housing with interchangeable leaf chambers. The CO2 analyzer is a differential, non-dispersive, infrared-type instrument calibrated for measurements of 0-1100 ppm.
The net exchange of CO2 between a leaf and the atmosphere is measured by enclosing a leaf section in the leaf-chamber and monitoring the exchange rate in CO2 concentration of air in the chamber during a short time interval of 20 to 30 seconds. Area of the leaf section in the chamber is measured and the net photosynthetic rate is calculated based on the rate of change in CO2, leaf area enclosed, volume of enclosure and air and leaf temperatures.
The measurements on photosynthesis and other parameters were made on a weekly basis after the start of treatments. The latest fully developed leaf (generally 3rd or 4th leaf from the top) was used for photosynthetic measurements. Measurements representing normal leaf position and orientation were replicated on four plants per lysimeter.
Chlorophyll measurements: Leaf chlorophyll measurements were made with a chlorophyll meter (Minolta SPAD 502 ). The SPAD-502 chlorophyll meter, is a nondestructive, hand held meter available for measurement of the green colour intensity in crop leaves which is directly related to leaf chlorophyll. The meter is light weight (225 g), powered by two AA alkaline batteries and can store up to 30 measurements.
The principle on which SPAD-502 works is based on the difference in light attenuation at wavelengths 430 and 750 nm. The 430 nm wavelength is in the near-infrared region, where no transmittance occurs. The ratio of the light transmittance at this wavelength, is processed by the instrument to produce a reading shown on a digital display. This reading is in SPAD (soil plant analysis development) units ranging from 0 to 80, which are values defined by Minolta to indicate the relative amount of chlorophyll contained in plant leaves.
The chlorophyll measurements were made on four plants per lysimeter (sixteen plants per treatment). The leaf chlorophyll measurements were made on weekly basis after the start of treatment.
RESULTS AND DISCUSSION
Photosynthetic rate: The statistical analysis of the data showed that, a week after WT treatments were established, photosynthetic rate for 0.15 m WT treatment was significantly lower than other treatments. Maximum rate was found for 0.45 m WT treatment (Fig. 2). Highest photosynthetic rates were observed for 0.30 m WT treatments on 44, 51, 58, 72 and 79 days after planting (DAP). However, on 65, 72, 86, 93 and 100 DAP, maximum values of photosynthesis were found for 0.60 m WT treatment. After reaching the peak rate (on 72 DAP), photosynthetic rates for 0.15 m WT treatment were found to be significantly lower than 0.60 m WT treatment. Differences between 0.30 and 0.45 m WT treatments were always non-significant, although the 0.45 m WT treatment at times gave higher photosynthetic rates than 0.30 m WT treatment. Also, photosynthetic rates for 0.45 and 0.60 m WT treatments were non significantly different except during the first and last week of the establishment of WT treatments. Highest and lowest photosynthetic rates achieved during the growing season were 20.15 (observed for 0.60 m WT treatment on 72 DAP) and 11.26 μmol/m2/s (observed for 0.15 m WT treatment on 44 DAP), respectively. For 0.15, 0.30, 0.45 and 0.60 m WT treatments, the photosynthetic rates varied from 11.26 to 14.52, 13.24 to 18.45, 13.82 to 19.39 and 12.06 to 20.15 μmol/m2/s, respectively.
Relationships were developed for photosynthesis as a function of DAP (Fig. 2). The results showed that the best fit models for all WT table treatments were quadratic. These models showed that a strong relationship between photosynthetic rate and DAP. The determination coefficients (R2) obtained for 0.15, 0.30, 0.45 and 0.60 m WT treatments were 0.72, 0.83, 0.86 and 0.92, respectively. This showed that DAP was better able to predict variation in photosynthetic rates if WT depth was lowered (to deeper depths). Under shallow WT depths, the crop was under stress due to excessive wetness, therefore, a lot of variation in mean photosynthesis was observed. This study provides an estimate of photosynthetic rate for different WT treatments as a function of DAP. These regression models need to be verified further with the data from field conditions before being used.
The ANOVA using the entire data showed that mean photosynthesis for 0.15, 0.30, 0.45 and 0.60 m WT treatments were 13.25, 15.94, 16.19 and 16.36 μmol/m2/s, respectively (Table 1). Although, the overall results showed a decrease in mean photosynthesis with the rise in WT depth, the differences between WT treatments were found to be statistically non-significant. The overall mean photosynthesis over the treatment period for all WT treatments was 15.44 μmol/m2/s.
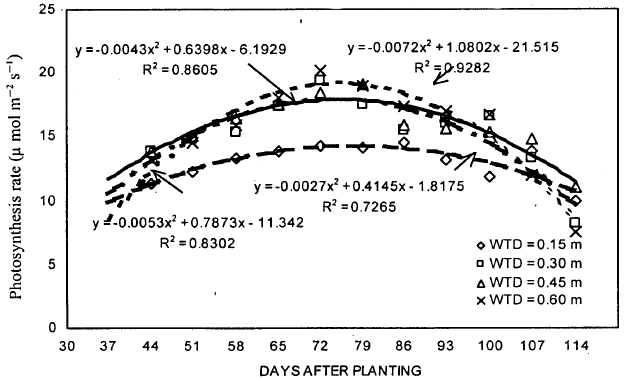 | |
| Fig. 2: | Photosynthesis rate of soybean during the growing season under different water table treatments |
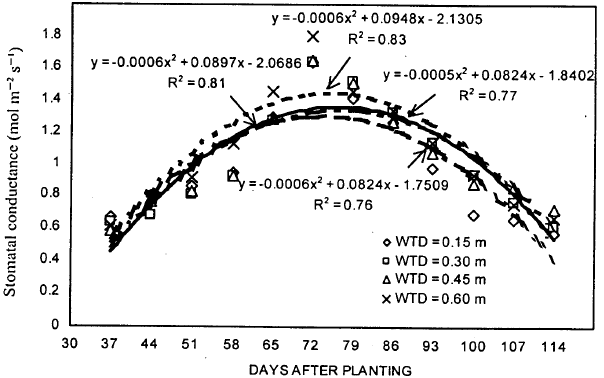 | |
| Fig. 3: | Stomatal conductance of soybean during the growing season under different water table treatments |
Stomatal conductance: The results showed quite inconsistency on stomatal conductance response to WT treatments (Fig. 3). A week after treatments were established, differences between WT treatments were found to be statistically significant. The stomatal conductance for 0.30 m WT treatment was significantly lower than 0.60 m WT treatment. For 44, 51, 58 and 64 DAP, stomatal conductance under 0.15 m WT treatment was higher than 0.30 and 0.45 m WT treatments. The stomatal conductance values for 0.30 and 0.45 m WT treatments were almost same on 51, 58, 65, 72 and 79 DAP. Highest stomatal conductance values for all WT treatments were achieved on 72 DAP. After reaching the peak, the trends were changed for different WT treatments and the stomatal conductance under 0.15 m WT treatment was lower than other three WT treatments. Also, stomatal conductance values obtained for 0.30 m WT treatment were higher than 0.45 m WT treatments. Although, the trends were inconsistent for 0.15, 0.30 and 0.45 m WT treatment, the stomatal conductance under 0.60 m WT treatment was always higher than other three WT treatments.
The overall mean stomatal conductance for 0.15, 0.30, 0.45 and 0.60 m WT treatments were 1.04, 1.10, 1.10 and 1.17 mol/m2/s (Table 1).
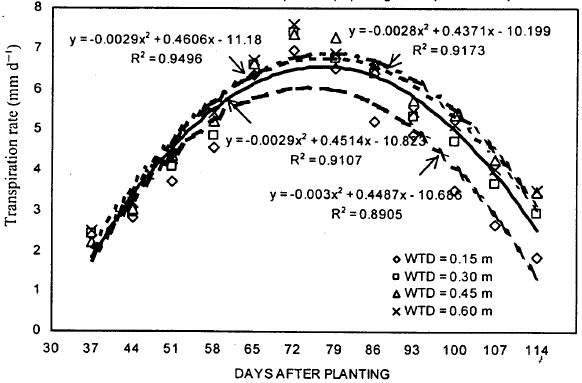 | |
| Fig. 4: | Transpiration rate of soybean during the growing season under different water table treatments |
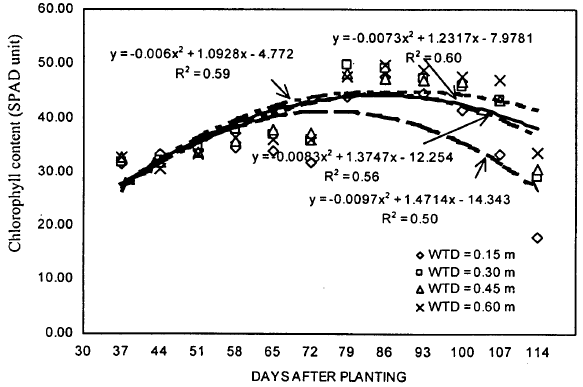 | |
| Fig. 5: | Chlorophyll content of soybean during the growing season under different water table treatments |
The mean stomatal conductance for all WT treatments during the treatment period was 1.10 mol/m2/s. Although, the stomatal conductance was lower under shallower WT depths, the differences in stomatal conductance between different WT treatments were non-significant.
The results from regression analysis showed that quadratic model was able to explain the variation in stomatal conductance better under different WT treatments (Fig. 3). The determination coefficients (R2) for 0.15, 0.30, 0.45 and 0.60 m WT treatments were 0.76, 0.81, 0.77 and 0.83, respectively. The stomatal conductance for all WT treatments increased with time until 72 DAP, thereafter, the values kept decreasing until last measurements were made. These trends are similar to those obtained for photosynthetic rate.
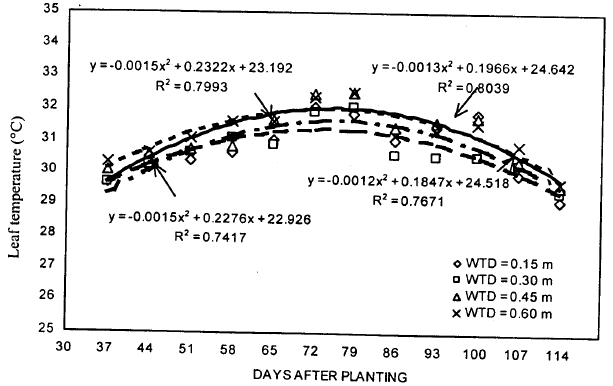 | |
| Fig. 6: | Leaf temperature of soybean during the growing season under different water table treatments |
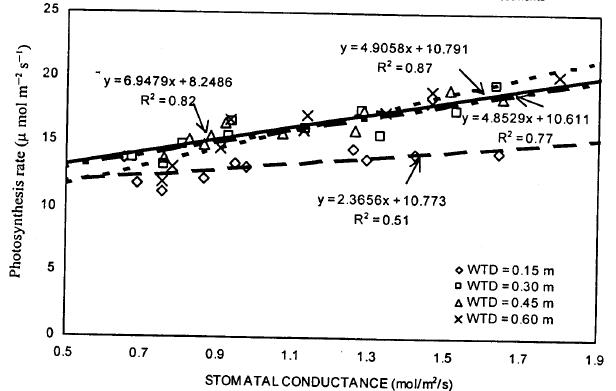 | |
| Fig. 7: | Relation between stomatal conductance and photosynthetic rate under different water table treatments |
Transpiration rate: The results showed that the transpiration rate was significantly affected by WT treatments (Fig. 4). A week after the WT treatments were established (on 44 DAP), transpiration rates for shallow WT depths were significantly lower than deeper WT depths. Canopies under 0.15 m WT treatments transpired 11% less water compared with the 0.60 m WT treatment. Highest transpiration rate on 44 DAP was found for 0.60 m WT treatment. These trends kept changing until peak transpiration rate were achieved on 72 DAP. Before reaching the peak, the highest transpiration rates were found either for 0.45 or 0.60 m WT treatment. From 86 DAP and afterward, the highest transpiration rates were achieved at 0.45 m WT treatment. The transpiration rates for 0.15 m WT treatment were always found to be lowest. The maximum transpiration rates of 6.93, 7.41, 7.35 and 7.60 mm/d were achieved for 0.15, 0.30, 0.45 and 0.60 m WT depths, respectively, on 72 DAP.
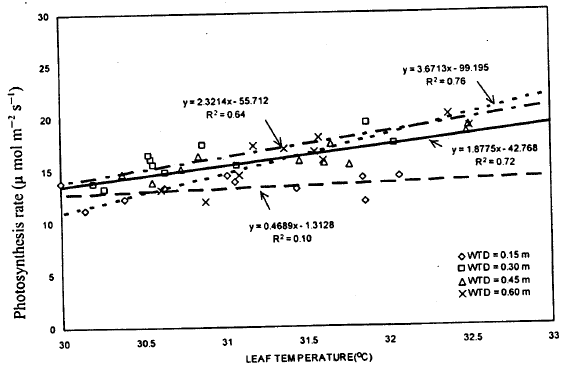 | |
| Fig. 8: | Relation between leaf temperature and photosynthetic rate |
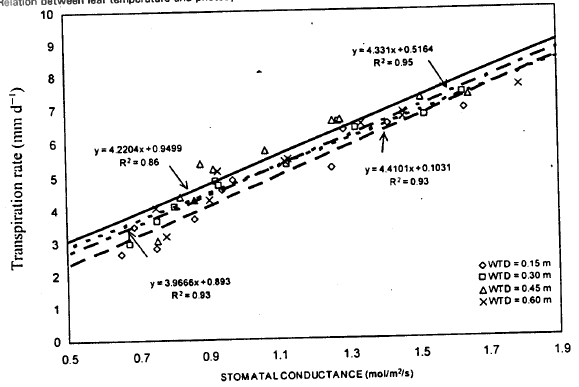 | |
| Fig. 9: | Relation between stomatal conductance and transpiration rate |
The lower transpiration rates at 0.15 m WT could be due to excessive water supply in the root zone which in turn caused stress, whereas, the slight reduction in transpiration rates at the 0.60 m WT depth could be due to inadequate moisture contents in the root zone.
These results are in agreement with the findings of Kalita and Kanwar (1992) and Mirjat (1994). They observed low transpiration rates of corn leaves at 0.20 and 1.1 m WT depths. Cannell and Jackson (1981) also reported that waterlogging causes shoot wilting in a very short time, which in turn causes physiological drought to the plant leaves by increasing resistance to water flow to the roots. When stomata are open wide enough transpiration rates are higher and in contrast, when roots reduce water supply to leaves, the plant leaves wilt and as a result, transpiration rate slow down significantly.
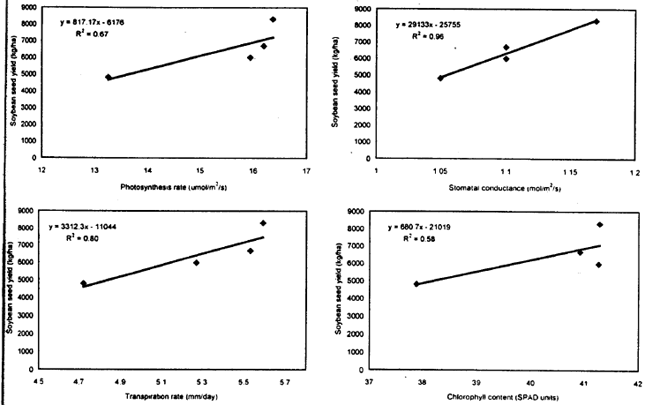 | |
| Fig. 10: | Relation between plant physiological parameters and soybean seed yield |
| Table 1: | Means of different plant physiological parameters using the data for all weeks of measurement |
 | |
1PHOTO - Photosynthetic rate; 2CON - Stomatal conductance; 3TRANS - Transpiration rate; 4CHLORO - Chlorophyll content; 5TLEAF - Leaf temperature; *LSD - least significant difference at the 0.05 probability level | |
| Table 2: | Analysis of variance for soybean yield (kg/ha) under different water table treatments |
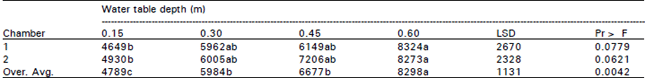 | |
| Means in rows followed by same letter are not significantly different at the 0.05 probability level | |
The overall analysis showed that transpiration rate was significantly different for all WT treatments (Table 1). The 0.45 m WT treatment gave the highest overall transpiration rate when compared with other WT treatments. Under 0.15 m WT treatment, leaves transpired 16% less water than those at 0.45 m WT treatment. Transpiration rates at 0.30 m WT depth were only 5% lower than 0.60 m WT depth.
The regression analysis results indicated that DAP was able to explain, on the average, 92% of the variation in transpiration rates (Fig. 4). The best-fit models obtained for different WT treatments were of second order polynomials (quadratic). The models predicted maximum transpiration rates of 6.07, 6.45, 6.65 and 6.94 for 0.15, 0.30, 0.45 and 0.60 m WT treatments respectively, on 72 DAP. The transpiration rate for all WT treatments were low early in the growing season, reaching to a peak on 72 DAP and declined thereafter. The models also predicted that transpiration rates for 0.45 m WT treatment were higher compared with other three WT treatments.
Chlorophyll content: Chlorophyll content of soybean leaves was determined weekly using chlorophyll meter (Fig. 5). Results indicated that, a week after WT establishment, the chlorophyll content for 0.15 m WT treatment was significantly higher than other three treatments. However, different trends were observed for rest of the growing season. During earlier part of the growing season (51, 58, 65, 72 and 79 DAP), maximum chlorophyll content was found either for 0.30 m or the 0.45 m WT treatment. For later part of the growing season (86, 93, 100 and 107 DAP), the highest values of chlorophyll content were found for 0.60 m WT treatment. Some consistency was observed during the last two weeks of treatments, where higher chlorophyll content was observed for deeper WT depths and lower was for shallow WT depths. Maximum chlorophyll content of 49.71 was achieved on 86 DAP under 0.60 m WT treatment. Chlorophyll content was lowest for 0.15 m WT treatment during most of the treatment period. Difference in chlorophyll contents between 0.30 and 0.45 m WT treatments and between 0.45 and 0.60 m WT treatments were always non-significant. For 0.15, 0.30, 0.45 and 0.60 m WT treatments, the mean values of chlorophyll content varied from 31.32 to 47.69, 31.96 to 49.79, 31.79 to 48.16 and 30.54 to 49.71, respectively.
The ANOVA showed an increase in chlorophyll content with the increase in WT depth (Table 1). The highest value of 41.28 was obtained for 0.60 m WT treatment and the lowest value of 37.88 was obtained for 0.15 m WT treatment. Although, the differences between WT treatments for chlorophyll content were statistically non-significant, the chlorophyll values for 0.15 m WT treatment were about 8% lower than 0.60 m WT treatment.
A second order polynomial curve was found to be the best-fit for chlorophyll data (Fig. 5). Although, the determination coefficients (R2) obtained were not very high (0.50, 0.56, 0.59 and 0.60 for 0.15, 0.30, 0.45 and 0.60 m WT treatments, respectively), consistent increase in their values indicate that better predictions could be obtained if higher WT depths are used. These curves indicate that chlorophyll content of soybean leaves is low early in the growing season, reach to peak value near the pod filling stage and declines thereafter. These trends are consistent with those obtained for photosynthesis and stomatal conductance.
Due to large inconsistency in results, definite conclusions can not be drawn about the effect of WT treatments on chlorophyll content of soybean leaves. However, it should be recognized that WT treatments in the present experiment were established for a period of 70 days as compared with other studies reported in literature where only short term effects were considered. Under prolonged shallow WT conditions, the effects are not clear until the later part of the growing season. For short term WT conditions (7 days of treatment), significant differences could be observed as achieved in this study also, a week after WT treatments were established.
Leaf temperature: The results showed that the leaf temperature values were lower during the early part of the treatment period, reaching to a peak and declined thereafter (Fig. 6). Highest values of leaf temperature were found for 0.60 m WT treatment on 79 DAP and the lowest values were observed for 0.15 m WT treatment on 107 DAP. A week after the WT treatments were established, leaf temperature was found to be increased with the increase in WT depth. Similar trends were obtained in the following week (on 51 DAP), however, there was some inconsistency in results afterwards. During the later part of the treatment period (on 86, 93, 100 and 107 DAP), lowest values of leaf temperature were obtained for 0.30 m WT treatment. The leaf temperature values for 0.60 m WT treatment were the highest during most of the WT treatment period. For 0.15, 0.30, 0.45 and 0.60 m WT treatments, the leaf temperatures during the treatment period ranged from 30.00 to 32.08, 30.20 to 32.05, 30.38 to 32.50 and 30.61 to 32.51oC, respectively. The leaf temperature values under different WT treatments were so close to each other that the separate analysis of all ten weeks of treatment period gave statistically non-significant results. Entire data was subjected to regression analysis to investigate the relation between leaf temperature and DAP (Fig. 6). These results revealed that a good correlation exists between leaf temperature and DAP. The determination coefficients (R2) for 0.15, 0.30, 0.45 and 0.60 m WT treatments were 0.74, 0.76, 0.80 and 0.80, respectively.
Similar trends have been reported in the literature. Allen et al. (1994) determined the midday leaf temperatures of soybean under drought stress in controlled-environment growth chambers. They found that leaf temperatures were non-significant during early stages of the water stress period for water stressed treatments. As water stress progressed, mid-day leaf temperature was also increased. For non-stressed treatments, an increase of >6oC (mostly late in the period) was observed in the leaf temperature under 330 ppm carbon environment.
Soybean yield: Crop yield data for the two growth chamber showed that the highest crop yields were obtained for 0.60 m WT treatment and the lowest was for 0.15 m WT treatment (Table 2). The ANOVA was performed using SAS procedures to determine the difference between yield means under four WT treatments for individual chambers. The results from growth chamber 1 showed that the average yield from 0.15 m WT treatment was significantly lower than other three WT treatments. The yield differences between 0.30, 0.45 and 0.60 m WT treatments were non-significant, though yields were always greater at lower WT depths. Similar trends were obtained from growth chamber 2 but the values of mean yield for 0.15, 0.30 and 0.45 m WT treatments were greater than chamber 1. Combined analysis of the two growth chambers improved the results and the yields for 0.60 m WT treatment were significantly greater than for 0.15, 0.30 and 0.45 m WT treatments. The average soybean yield obtained for 0.15 m WT treatment was 20, 28 and 42% lower than mean yield for 0.30, 0.45 and 0.60 m WT treatments, respectively. The yield values for 0.30 m WT treatment were 10 and 28% lower than yield for 0.45 and 0.60 m WT treatments, respectively. The overall mean yield for 0.45 m WT treatment was about 20% lower than 0.60 m WT treatment but only 10% higher than mean yield for 0.30 m WT treatment. The differences between the 0.30 and 0.45 m WT treatments were non-significant.
The soybean yield for different WT treatments obtained from this experiment are greater than those obtained in field experiments (Cooper et al., 1991; Cooper et al., 1992; Oosterhuis et al., 1990). The reason for these greater yields could be the effect of a controlled environment that removed stresses like very dry or very wet climatic conditions.
Photosynthesis and stomatal conductance: Relationships between photosynthetic rates and stomatal conductance were developed through linear regression analysis (Fig. 7). Stomatal conductance values were the good predictor of photosynthetic rate, accounting for, on the average 74% of the variation in photosynthetic rates. The high values of determination coefficient (R2) for all WT treatments indicated that the linear models were appropriate to explain the relationships between these parameters and were fitted well to their respective data sets. Comparison between WT treatments based on R 2 values showed that the 0.45 m WT treatment yielded higher R2 values than did 0.15, 0.30 and 0.60 m WT depths. This provided the evidence that adequate supply of water to the root zone accelerated stomatal conductance, which in turn increased the photosynthetic rate. The lower values of R2 with 0.15 m WT depth, might explain that excessive water stress did affect the stomatal conductance, which reduced the photosynthetic rate.
Control of stomatal opening and closure is important to photosynthesis and its response to environmental stresses. The decrease in photosynthesis during the water stress period can be due to either non stomatal or stomatal effects (Cox and Jolliff, 1987; Wong et al., 1985; Boyer and Bowen, 1970). Cox and Jolliff (1987) reported that carbon dioxide exchange rate (CER) of soybean was reduced up to 50% with the increase in stomatal resistance due to water stress. They suggested that stomatal closure was responsible for reduced CER in dry land soybean. Stomatal movements provide the leaf with opportunity to change the CO2 both between atmosphere and the site of carboxylation and the rate of transpiration (Farquhar and Sharkey, 1982). Mohanty and Boyer (1976) reported that at low leaf water potential, photosynthesis can be reduced by stomatal closure. Stomatal closure due to water stress decreased leaf transpiration, which in turn, increased leaf temperature and decreased photosynthesis (Ehrler et al., 1978).
Photosynthesis and leaf temperature: Optimum leaf temperature is important for photosynthesis. Low leaf temperature reduced assimilation rate by reducing activity of rubisco and the capacity for electron transport, while high leaf temperature also reduced photosynthetic rate by reducing electron transport capacity and increasing the rate of CO2 evolution (Farquhar and Sharkey, 1982). The regression analysis was performed to investigate the relationship between leaf temperature and photosynthetic rate (Fig. 8). The result showed that the leaf temperature, on the average, was able to explain 56% variation in photosynthetic rate. Good correlation was found for all WT treatments except 0.15 m WT treatment (R2= 0.10). Such a low determination coefficient for 0.15 m WT treatment indicated that relationship between photosynthetic rate and leaf temperature does not hold good for very shallow WT conditions.
Transpiration rate and stomatal conductance: Transpiration rate is controlled by stomatal movements. Stomatal closure due to water stress decreased leaf transpiration, which in turn, increased leaf temperature and decreased photosynthesis (Ehrler et al., 1978). Relationship between transpiration and stomatal conductance for different WT treatments was investigated (Fig. 9). A strong positive correlation between transpiration rate and stomatal conductance for different WT treatments was observed. The stomatal conductance was found to be a good predictor of transpiration rate, accounting for 92% variation in transpiration rate. The high values for R2 for all WT depths indicated that the linear models were appropriate to explain the relationships between these parameters. Comparison between WT treatments based on R2 values showed that 0.30 m WT depth yielded higher R2 values than other three WT treatments.
Relation between plant physiological parameters and soybean seed yield: Relationships were developed between photosynthetic rate, stomatal conductance, transpiration rate, chlorophyll content and seed yield of soybean (Fig. 10). These figures showed a linear increase in soybean seed yield with the increase in photosynthetic rate, stomatal conductance, transpiration rate and chlorophyll content. Stomatal conductance and transpiration rate were found to be good predictors of soybean seed yield, accounting for 96 and 80% of the variation in seed yield, respectively. Although, the photosynthetic rate was able to express only 67% of the variability in seed yield, the seed yield was found to be increased with the increase in photosynthetic rate. The chlorophyll content was not observed as a good predictor of soybean yield.
Physiological responses of soybean to different WT treatments were determined in environmentally controlled chambers. Photosynthesis and stomatal conductance were significantly affected by shallow water tables after a week of WT establishment. Transpiration rate was the single parameter found to be highly responsive to WT depths. Shallow WT depths significantly reduced transpiration rates of soybean leaves compared with deeper WT depths. Effect of WT treatments on chlorophyll content of leaves was not observed during early part of the growth period, however, the results of last two weeks of measurements showed significant differences between different WT treatments for these two parameters. Shallow water table depths significantly reduced the plant height and grain yield compared with deeper WT depths. Highest soybean yield was obtained under 0.60 m WT depth and the lowest was for 0.15 m WT depth. Average yield obtained at 0.15 m WT was 48% lower than 0.60 m WT treatment. It seems evident from the results of this study that a strong positive relation exist between leaf temperature and photosynthesis and between transpiration rates and stomatal conductance. Seed yield was found to be increased with the increase in photosynthesis, stomatal conductance and transpiration rate.
REFERENCES
- Allen, Jr. L.H., R.R. Valle, J.W. Mishoe and J.W. Jones, 1994. Soybean leaf gas-exchange responses to carbon dioxide and water stress. Agron. J., 86: 625-636.
Direct Link - Boyer, J.S. and B.L. Bowen, 1970. Inhibition of oxygen evolution in chloroplasts isolated from leaves with low water potentials. Plant Physiol., 45: 612-615.
Direct Link - Cooper, R.L., N.R. Fausey and J.G. Streeter, 1991. Yield potential of soybean grown under a subirrigation/drainage water management system. Agron. J., 83: 884-887.
CrossRefDirect Link - Cooper, R.L., N.R. Fausey and J.G. Streeter, 1992. Effect of water table level on the yield of soybean grown under subirrigation/drainage. J. Prod. Agric., 5: 180-184.
CrossRefDirect Link - Cox, W.J. and G.D. Jolliff, 1987. Crop-water relations of sunflower and soybean under irrigated and dryland conditions. Crop Sci., 27: 553-557.
CrossRefDirect Link - Ehrler, W.L., S.B. Idso, R.D. Jackson and R.J. Regon, 1978. Diurnal changes in plant water potential and canopy temperature of wheat as affected by drought. Agron. J., 70: 999-1004.
CrossRefDirect Link - Farquhar, G.D. and T.D. Sharkey, 1982. Stomatal conductance and photosynthesis. Ann. Rev. Plant Physiol., 33: 317-345.
CrossRefDirect Link - Hesketh, J.D., 1963. Limitations to photosynthesis responsible for differences among species. Crop Sci., 3: 493-496.
Direct Link - Huber, S.C., H.H. Rogers and F.L. Mowry, 1984. Effects of water stress on photosynthesis and carbon partitioning in soybean (Glycine max L. Merr.) plants grown in the field at different CO2 levels. Plant Physiol., 76: 224-249.
PubMed - Kariya, K. and S. Tsunoda, 1972. Relationship of chlorophyll content, chloroplast area index and leaf photosynthesis rate in Brassica. Tohoku J. Agric. Res., 23: 1-14.
Direct Link - Kalita, P.K. and R.S. Kanwar, 1992. Energy balance concept in the evaluation of water table management effects on corn growth: Experimental investigation. Water Resour. Res., 26: 2753-2764.
CrossRefDirect Link - Keck, R.W. and J.S. Boyer, 1974. Chloroplast response to low leaf water potentials. III. Differing inhibition of electron transport and photophosphorylation. Plant Physiol., 53: 474-479.
CrossRefDirect Link - Nicolodi, C., A. Massacci and G. Di Marco, 1988. Water status effects on net photosynthesis in field-grown alfalfa. Crop Sci., 28: 944-948.
CrossRefDirect Link - Oosterhuis, D.M., H.D. Scott, R.E. Hampton and S.D. Wullschleger, 1990. Physiological responses of two soybean [Glycine max (L.) Merr] cultivars to short-term flooding. Environ. Exp. Bot., 30: 85-92.
CrossRefDirect Link - Reicosky, D.C., R.J. Millington, A. Klute and D.B. Peters, 1972. Patterns of water uptake and root distribution of soybeans (Glycine max L.) in the presence of a water table. Agron J., 64: 292-297.
CrossRefDirect Link - Sarwar, T. and R.S. Kanwar, 1996. NO3-N and metolachlor concentrations in the soil water as affected by water table depth. Trans. ASAE., 39: 2119-2129.
CrossRefDirect Link - Wong, S.C., I.R. Cowan and G.D. Farquhar, 1985. Leaf conductance in relation to rate of CO2 assimilation. III. Influences of water stress and photo inhibition. Plant Physiol., 78: 830-834.
Direct Link








