Research Article
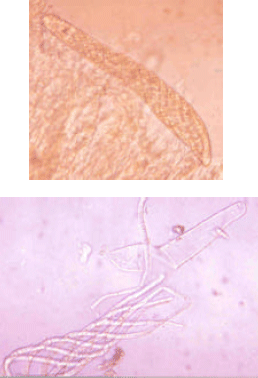
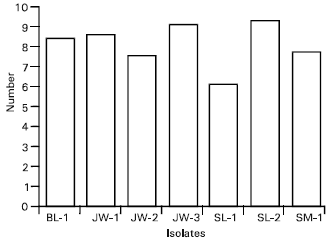
Morphological and Pathological Variations in the Indonesian Cochliobolus heterostrophus (Pleosporaceae, Pleosporales, Euascomycetes)
Department of Plant Protection, University of Lampung, Bandar Lampung 35145, Indonesia
Subli Mujim
Department of Plant Protection, University of Lampung, Bandar Lampung 35145, Indonesia
Titik Nur Aeny
Department of Plant Protection, University of Lampung, Bandar Lampung 35145, Indonesia