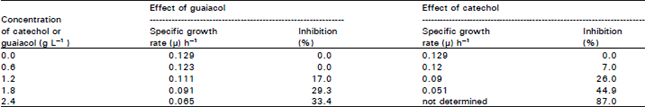
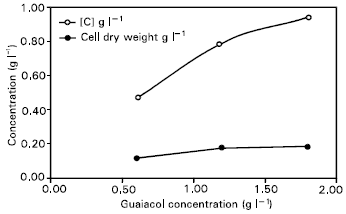
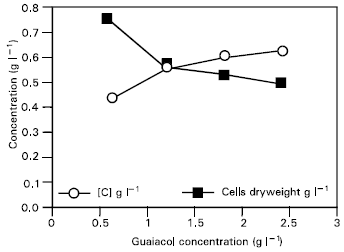
Research Article
Catechol Synthesis via Demethylation of Guaiacol by Anaerobic Bacterium Acetobacterium woodii DSM 1030
Department of Chemical and Process Engineering, Faculty of Engineering, Universiti Kebangsaan Malaysia 43600 UKM Bangi Selangor Malaysia
Zainatul Asyiqin
Department of Chemical and Process Engineering, Faculty of Engineering, Universiti Kebangsaan Malaysia 43600 UKM Bangi Selangor Malaysia
Muhammad Zaki
Department of Chemical and Process Engineering, Faculty of Engineering, Universiti Kebangsaan Malaysia 43600 UKM Bangi Selangor Malaysia