Research Article
Mutagenic Potential of Imazethapyr Herbicide (Pursuit*) on Vicia faba in the Presence of Urea Fertilizer
Biological Sciences and Geology Department, Faculty of Education, Ain Shams University, Cairo, Egypt
Herbicides are widely used on the global base in order to minimize the loss in economic crops due to plant competition. Number of investigators had studied the side effect of the herbicides on the hereditary material of different plant cells. They found that these chemicals had genotoxic effects on both mitotic and meiotic cells when treated with high doses (El-Abidin Salam et al., 1993; Burroughs et al., 1996). In spite of that the herbicides have been used extensively to increase crop productivity. Little is known about their effect on hereditery material in the presence of different fertilizers. Nitrogen is a vitally important plant nutrient, the supply of which can be controlled by man. Plants normally contain 1 and 5% by weight of this nutrient. It is absorbed by plants in the form of nitrate and ammonium ions and as urea. In moist, warm and well-aerated soils the NO form is dominant (Tisdale et al., 1985). Recent studies have been consolidated by exploiting the interaction between extra nutrients supplied by fetilizers, and herbicides. Such interaction may lead to increase in the yield. The effect of some N-fertilizers on mitotic index, cell division and chromosomal aberrations was studied by El-Awady (1996). She studied 3 nitrogenous fertilizers: Urea, calcium-nitrate and ammonium sulfate on Allium cepa and she reported that the three fertilizers did not alter processes regulating the entry of cells into mitosis. The use of N-fertilizer combined with pesticide application either reversed the effect of the pesticide (Reynolds, 1986) or did not reduce its effect Vintila et al. (1989). Meanwhile El-Awady (1996) reported that the interaction between urea and fungicide hinosan had an antagonizing effect on the mitotic activities. Electrophoretic techniques of protein have been used as a successful tool to estimate the possible mutagenic potentialities produced due to continuous and accumulative pollution by chemicals and pesticides and correlate the produced variation with chromosomal aberrations caused by these environmental pollutants (Gazaly, 1994).
Therefore, the present investigation was planned to study the mutagenic potentialities of the herbicide imazethapyr (pursuit) singly and its combination with the N-fertilizer urea as revealed by 2 approaches using faba beans:
| 1. | Cytological analysis of mitotic and meiotic divisions, chromosomal aberrations and its significance |
| 2. | Biochemical analysis using electrophoretic banding pattern on M2 seed storage proteins |
Seeds: Seeds of pure strains of Vida faba (cv. Giza 3) were obtained from the Crop Research Institute, Agricultural Research Center (ARC), Giza, Egypt.
Chemicals: The herbicide imazethapyr (pursuit*) and the nitrogenous fertilizer (urea) have been kindly supplied by the General Administration of Pesticides, Ministry of Agriculture of Egypt.
Cytological procedures: For pursuit herbicide, the sublethal and the threshold concentrations were determined, then four concentrations intermediate between these two limiting concentrations were selected for further experiments. These are, 12.5, 6.25, 3.12 and 1.56 ml/L. The concentration 0.9 g/L urea was used in this study.
For mitotic analysis, two sets of rooted faba beans (1.5-2 cm radicle length) were used as follows: first set were placed in different concentrations of the herbicide mentioned above in addition to the control (roots immersed in tap water, -ve control). The second set of faba bean roots were immersed in the previous four concentrations of pursuit to which 0.9 g/L urea fertilizer was added, in addition to some immersed in urea (+ve control). Two-exposure time 4 and 24 hours for both sets were approached. Then, after each treatment, the root tips were cut-off and fixed in Carnoy's (3:1) ethanol:glacial acetic acid. Cytological preparations were carried out using the Feulgen squash technique. In cells undergoing mitotic division, observed abnormalities as well as their total percentages were scored. Five replicates were selected for each concentration and the controls. The data obtained from the different treatments were statistically analyzed using the t-test.
For meiotic analysis, faba beans plants were sprayed once at the flowering stage (6 weeks old) with the four concentrations of the herbicide singly and combined with urea. Control plants were sprayed with distilled water (-ve control) and urea (+ve control). Ten flower buds from ten different plants were collected at random 24 hours and 15 days after spraying with each concentration of the herbicide singly and combined as well as controls. Then flower buds were fixed in (6:3:1) ethanol:glacial acetic acid:chloroform, stained and smeared using aceto carmine method then examined, and chromosome aberrations in the PMC's scored.
Biochemical analysis: SDS-polyacrylamide gel electrophoresis procedure was carried out according to Laemmli (1970). The molecular weight of the resulting protein banding patterns were estimated according to Matta et al. (1981). The dry M2 seeds of Vicia faba plants whose parents were previously sprayed with the herbicide either singly or combined with urea were decoated and milled to fine powder. Total proteins were extracted overnight using 0.2 M Tris/HCL buffer, pH 6.8 containing 2% SOS. Centrifugation was carried out at 10.000 rpm for 10 minutes, then 30 μ L supernatant were loaded in 12.5% acrylamide slab gels containing 10.5 SIDS, run at 15 mA for about 30 minutes, followed by 25 mA for 6-7 hours. The protein bands were visualized by staining the gels with coomassie brilliant blue (COBB, R-250) for 24 hours. Alteration in the protein profiles was scored as presence or absence and as decrease or increase in band intensity. Gel was analyzed using SDS (Gel Documentation System) computer programe.
Cytological analysis: Table 1 and 2 show the effect of the herbicide singly as well as combined with the fertilizer (respectively) on the mitotic index and induction of chromosome aberrations. It is clear that mitotic index value progressively decrease with both duration of treatment and concentrations used. Such mitodepressive effect was more pronoouced after treatment with pursuit in a single form than those exerted by its combination with urea. It is noteworthy that the apparent increase of mean values of mitotic index in the different concentrations of the combination of pursuit with urea (Table 2) is in fact due to the higher mean value of positive control than that of negative control (Table 1). Mitotic activity reached minimum values 0.98% and 2.13% after 24 hours treatment with the highest concentration of either the herbicide singly or combined with urea, respectively. Such decrease in the mitotic index was found to be statistically highly significant at higher concentrations and significant at the rest of concentrations, but at (1.56 mI/L) no significant effect after 4 hours of treatment was detected.
The reduction of mitotic activity seems to be a common effect of most herbicides tested for their action on mitosis. Many herbicides were reported by a number of authors to exert this effect such as rancho (Sobhi and Haliem, 1990), pendimethalin (Barakat and Hassan, 1997) and atcanex (Ghareeb, 1998). Such mitotic inhibition has been attributed to a number of factors (Deysson, 1968). Among these are: the blocking of mitotic cycle during interphase (Mohandas and Grant, 1972), on the basis of chromosome aberrations and/or altered metabolic rate (Polson and Adams, 1978); the interference of the herbicide with the normal process of mitosis by reducing the number of the dividing cells (El-Khodary et al., 1989), the inhibition of certain types of nuclear protein synthesis essential in the cell cycle (Kim and Bendixen, 1987). Also the reduction in the mitotic rate may be due to the inhibitory effect of herbicides during the formation of various metabolites necessary for normal sequence of mitosis (Howell, 1998; Ghareeb, 1998). Other investigators have related the depression in mitotic index values to the inhibition of DNA synthesis (Beu et al., 1978).
Pursuit herbicide had a great effect on the normal behavior of chromosomes during mitosis. It induced different types of abnormalities. Their frequency depends on the concentration and duration of treatment i.e. increasing concentration and duration of treatment led to parallel increase in the frequencies of chromosome abnormalities. It reached a maximum value of 55.63% after 24 hours with the highest concentration (12.5 ml/L) of the herbicide. Statistical analysis revealed that, after 4 and 24 hours, all the applied concentrations were effective in inducing either a non significant or significant besides highly significant increase as shown in Table 1.
Armbruster et al. (1991) as well as others reported that the induction of mitotic abnormalities was a common effect of most herbicides tested for their action. Concerning the interaction of pursuit with urea, Table 2 shows that, induced abnormalities observed in roots treated with pursuit in the absence of urea still appeared after combined treatment for both 4 and 24 hours. It reached a maximum value of 22.11% after 4 hours with 12.5 mill herbicide combined with the fertilizer. Also, it was found to be significant at most of the conducted treatments. The total frequencies of abnormalities increased with concentration but decreased by increasing duration. The recorded values of abnormalities revealed a considerable decrease as compared with those recorded after treatment with pursuit singly (Table 2). Chromosome aberrations, including stickiness, C-metaphase, laggards, bridges, disturbed phases and breaks were observed (Fig. 1-4). Stickiness being the most pronounced phenomenon reaching the maximum value of 31.11 and 11.20% at metaphase and ariatelophase respectively following the 24 hours treatment in 12.5 ml/L herbicide (Table 1). Patil and Bhat (1992) suggested that, stickiness is a type of physical adhesion involving mainly the proteinacious matrix of chromatin material.
C-metaphase was recorded in considerable frequency after the different treatments with all concentrations, reaching a maximum value of 20.30% at 12.5 mI/L pursuit (Table 1). This aberration is due to the inhibition of spindle fibers (Deysson, 1968).
Bridges were observed at the ana-telophase nearly in all treatments when the herbicide was used singly. When combined with urea this aberration was found in most treatment after 4 hours, whereas after 24 hours it appeared only in the highest concentration as shown in Table 2. The formation of bridges could be attributed to chromosomal stickiness or to chromosome breakage and reunion (Tomkins and Grant, 1972).
Among aberrations that appeared frequently was laggards. The induction of which could be attributed to the failure of the spindle apparatus to organize and functions in a normal way (Patil and Bhat, 1992). Disturbance appeared at metaphase and ana-telophases in single or combined treatments of herbicide showing the highest values of 14.11 and 11.11% at metaphase following 4 hours treatment. Other aberrations (multipolar and breaks) appeared in variable percentages. The effect of different treatments with the herbicide singly or combined on PMC's of Vicia faba following 24 hours and 15 days after spraying are summarized in Table 3 and 4 where different types of chromosomal abnormalities covering the 1st and 2nd meiotic stages were scored. Data revealed a pronounced decrease in the meiotic irregularities following combined treatments as compared with values after treatment with the herbicide singly. For both treatments (single and combined) the total % of abnormalities in PMC's increased with increasing concentration in both meiotic divisions for 24 hours except at 3.12 mIlL. The same trend was observed after 15 days for the 1st meiotic division, whereas in the 2nd meiotic division, no particular trend can be deduced. The highest values of meiotic abnormalities (22.16 and 6.20%) were recorded 15 days after spraying with 12.5 ml/L, pursuit singly at both meiotic divisions respectively as evident from Table 3. This means that 15 days are not long enough for recovery. These percentages of abnormalities were reduced in case of the combination of the herbicide with urea at 1st and 2nd meiotic divisions (Table 3). Thus urea seems to minimize the effect of the herbicide. With respect to the increase of abnormalities with increasing concentration, similar findings were ovserved by El-Abidin Salam et al. (1993) and Hassan and Barakat (1998).
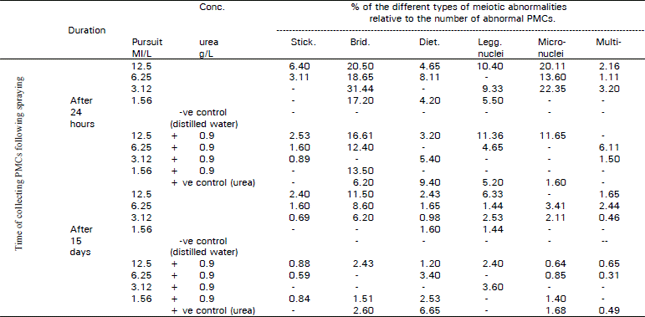
Regarding types of aberrations stickiness, laggards, disturbance, and micro and multinuolei were observed (Table 4 and Fig. 5-10).
| Table 1: | Mean of mitotic index, mean percentage of abnormal mitosis and frequencies of different types of metaphase and aria- telophase abnormalities after treating Vide tab: root tips with different concentrations of pursuit herbicide for 4 and 24 hours |
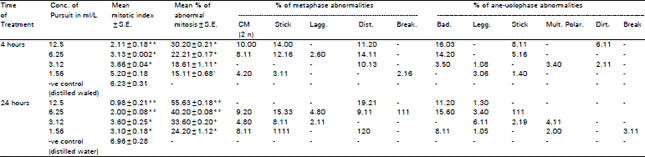 | |
| ** Significant from control at 0.01 level (t-test), *Significant from control at 0.05 level (t-test) | |
| Table 2: | Mean of mitotic index, mean percentage of abnormal mitosis and frequencies of different types of metaphase and ans. telophase abnormalities after treating Weis fib root tips with different concentrations of pursuit herbicide combined with 10.9 WU urea fertilizer for 4 and 24 hours |
 | |
| *Significant from control at 0.05 level (t-test), **Significant from control at 0.01 level (t-test) | |
| Table 3: | Mean total percentage of abnormal PMC's in the first and second meiotic divisions and percentage of abnormal PCM's in each phase after 24 hours and 15 days preying Vieie faba plants with pursuit herbicide singly and with combination of (0.9 g/L) urea fertilizer |
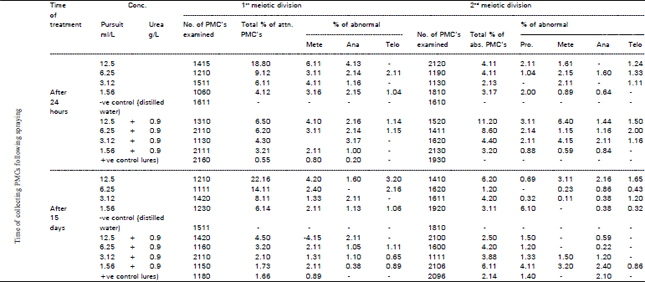 | |
| *Significant from control at 0.05 level (t-test)**Significant from control at 0.01 level (t-test) | |
Concerning micronuclei the highest value of (22.35%) was observed in samples collected after 24 hours of spraying with the herbicide (3.12 ml/L). This value dropped to 2.11% after 15 days: when combined with urea this aberration disappeared 24 hours and 15 days following spraying, this may be attributed to the effect of urea in counteracting the mutagenic effect of the herbicide. It is well known that micronuclei are true mutagenic aspects which may lead to a loss of genetic material. This mutagenic effect was estimated as a percentage of micronouclei formed in interphase (Burroughs et al., 1996).
Multinucleated cells are illustrated in Fig. 10, their frequency as presented in Table 4 showed the same trend being higher after 24 hours treatment and reaching a minimum value after 15 days.
 | |
| Fig. 1-4: | Mitotic abnormalities induced by treating Vicia faba root tips with pursuit for 4 hours (at X800). 1, sticky metaphase, 12.5 ml/L. 2, metaphase with chromosome breakage, 6.25 ml/L. 3, disturbed anaphase with double bridges, 12.5 ml/L. 4, anahphase with lagging chromosome and bridges, 3.12 ml/L |
 | |
| Fig. 5-10: | Meiotic abnormalities induced by spraying Vica faba plant with 12.5 ml/L. Pursuit, 24 hours after spraying (at X2000). 5, stickey metaphasae I with two lagging bivalents. 6, disturbed sticky anaphase I with bridges and lagging chromosome. 7, stickey metaphase II with laggards and fragments. 8, distrubed anaphase II with bridges and laggard. 9, anaphase II with laggard and break, 10, late telephase II with micro and multinuclei |
| Table 4: | Percentages of abnormal meiotic types occurring in PMCs, 24 hours and 15 days after spraying Vicia faba with pursuit herbicide singly and with combination of 0.9 g/L urea fertilizer |
 | |
 | |
| Fig. 11: | Electrophotograph produced by denatured PAGe analysis of general protein patterns of M2 seeds of Vida faba plants whose parents were treated with different concentrations of pursuit in non-combined and combined (with 0.9 g/L urea) forms Lane M represents the molecular weight marker. Lanes 1,2,3 and 4 represent samples treated with 12.5, 6.25, 3.12 and 1.56 ml/L. of pursuit respectively. Lanes 5,6,7 and S represent samples treated with doses of pursuit combined with 0.9 g/L urea fertilizer. Lanes 9 and 10 represent samples treated with urea and distilled water as (+ve) and (-ve) controls respectively. ➤ head arrows indicate the disappearance of band in lanes 1-6. Open arrow indicates the new bands formed in the same land. |
Biochemcial analysis: The obtained data revealed that the tested herbicide in non-combined and combined forms caused many changes in the protein banding patterns of Vicia faba M2 seeds whose parents were previously treated with both forms (Table 5, Fig. 11 ).
The total number of bands is 30 bands with molecular weights ranging between 132.11-18.40 kDa. The highest number of bands (19) are recorded in the non-treated samples (+ve and -ye controls). The recorded changes were expressed as variations in the number of separate bands, disappearance or appearance of certain bands, difference in the intensity of one or more protein polypeptides and in bands relative mobilities. Similar results were obtained by other authors following different treatments (Badr et al., 1983; EI-Awady, 1996; Abdelsalam et al., 1997). Figure 11 shows the disappearance of some bands (indicated by head arrows) in lanes 1-6 as compared with the control (either+ve or -ve) in lanes 9 and 10. The disappearance of these bands could be attributed to the loss of the genetic material, this is in agreement with Barakat and Hassan (1997). New bands appeared at approximately molecular weight 53 kDa at the same lanes (indicated by open arrow as shown in Fig. 11). This may be explained on the basis of mutational event at the regulatory system of an unexpected gene (s) that activate it (Abdelsalam et al., 1997).
The changes in band intensity could be explained on the basis of induction of gene mutation at the regulatory system which modulates, attenuates or increases transcription rate of a particular structural gene (Grist et al., 1992). According to El-Abidin Salam et al. (1993) the recorded changes in band intensity could be attributed to the cytological abnormalities.
Changes in bands relative mobilities could be attributed to the occurrence of point mutation in the concerned structural genes that create stop codon prior or post the original. This gave rise to the production of shorter or longer polypeptide chains as reported by Barakat and Hassan (1997).
The above mentioned variations in the protein patterns of the differently treated plants might be assumed to result from changes in the gene expression that could be transferred to the M2 seeds. This assumption excluded that protein variability might be attributed to seed heterogeneity or cross-pollination since a pure broad bean line was used and the experimental conditions were thoroughly controlled.
| Table 5: | Comparative analysis of relative concentration (band %) mobility rate (Rm) and molecular weight (M.Wt) of the different types of protein bands of M2 seeds of Vicia faba plants whose parents were treated with different concentrations of pursuit herbicide in the presence or in absence of urea fertilizer |
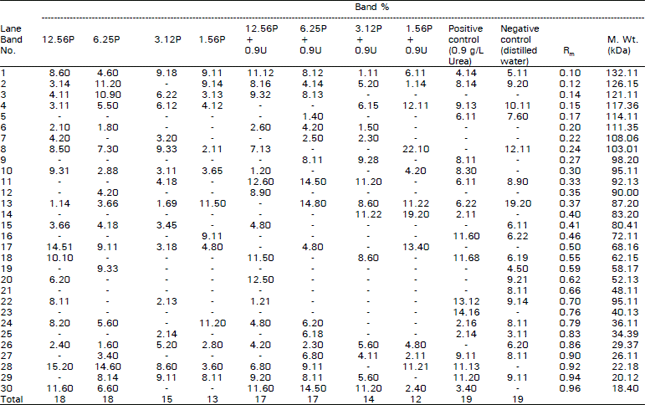 | |
| Pursuit herbicide (P) in mg/L. Urea frertilizer (U) in g/L | |
Events causing changes in the gene expression do mainly occur at transcriptional levels, but control can also be exercised at a post-transcriptional, translational or even post-translational stage (Anderson and Beardall, 1991; Battey et al., 1993; Hopkins, 1995).
Thus, the different treatments applied in the present work are presumed to affect the protein patterns at one or more of the above mentioned control points. Since the results obtained from protein analysis are in harmony with those obtained from chromosomal studies (based on data from mitotic and meiotic divisions), we may conclude that urea fertilizer plays a role in decreasing the harmful effect of the herbicide.