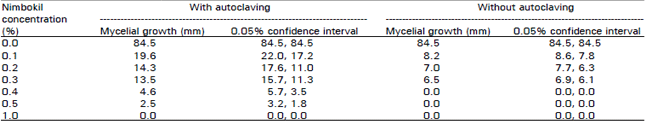
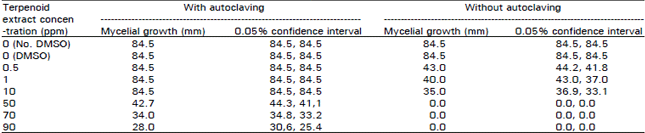
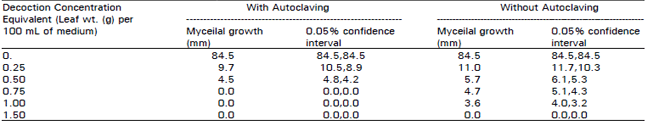
Research Article
In Vitro Antifungal Activity of Neem Products Against Phytophthora Infestans
Crop Diseases Research Institute, NARC, Pakistan Agricultural Research Council P.O. Box 1081, Islamabad
Shazia Hameed
Crop Diseases Research Institute, NARC, Pakistan Agricultural Research Council P.O. Box 1081, Islamabad
Iftikhar Ahmad
Crop Diseases Research Institute, NARC, Pakistan Agricultural Research Council P.O. Box 1081, Islamabad
Najma Ayub
Biology Department, Quaid-e-Azam University, Islamabad, Pakistan
R.H. C. Strang
Biochemistry and Molecular Biology Division, University of Glasgow, Glasgow, G128 QQ, Scotland, UK