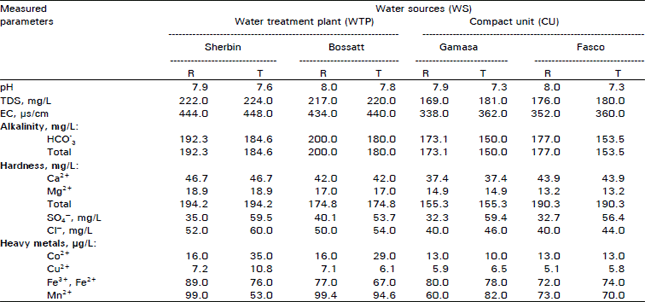
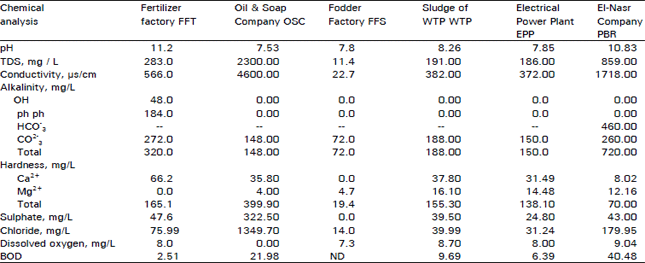
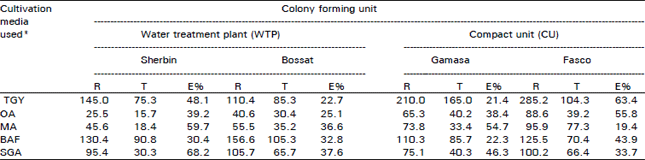
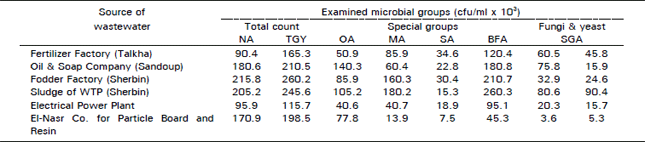
Research Article
Chemical and Microbiological Analyses of Certain Water Sources and Industrial Wastewater Samples in Egypt
Department Microbiology, Faculty of Agriculture, Mansoura University, Mansoura, Egypt
M.M. El-Defrawy
Department of Chemistry, Faculty of Science, Mansoura University, Mansoura, Egypt
F. El-Zawawy
Department of Chemistry, Faculty of Science, Ain-Shams University., Cairo, Egypt
D. Makia
Department of Chemistry, Faculty of Science, Ain-Shams University., Cairo, Egypt