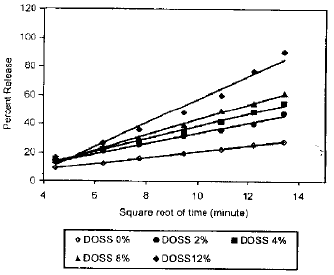
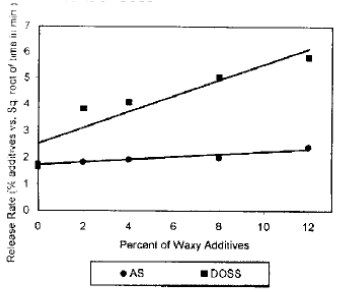
Research Article
Preparation and Stability Study of Diclofenac Sodium Suppositories
Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
R. Jalil
Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
M. A. Islam
Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh
S. M. Shaheen
Department of Pharmacy, University of Rajshahi, Rajshahi-6205, Bangladesh