Research Article
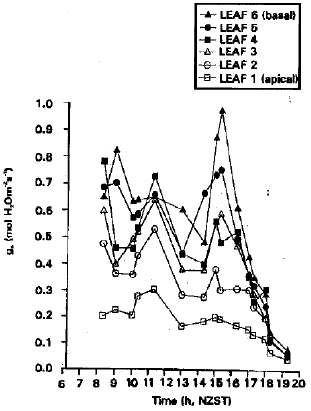
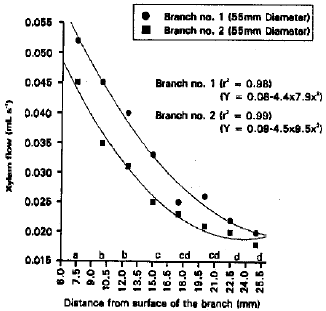
To Study Stomatal Conductance at Different Leaf Positions and Xylem Flow Rate at Different Depths in the Apple Branch
Horticulture, Agricultural Research Institute, Tarnab, Peshawar, NWFP, Pakistan
M. C. Trought
Soil, Plant and Ecological Sciences Division Lincoln University, Canterbury, New Zealand
R. Noor
Horticulture, Agricultural Research Institute, Tarnab, Peshawar, NWFP, Pakistan
A. Samad
Horticulture, Agricultural Research Institute, Tarnab, Peshawar, NWFP, Pakistan