Research Article
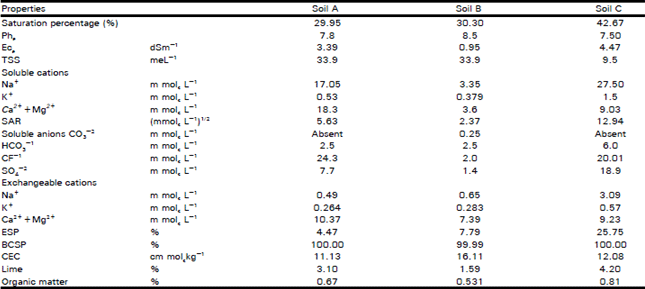
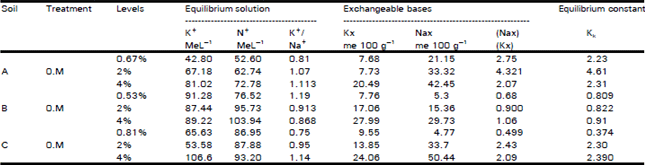
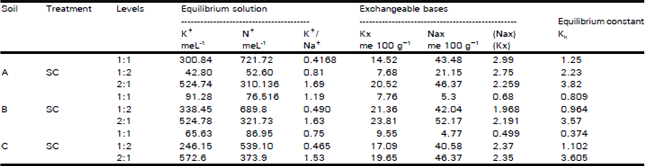
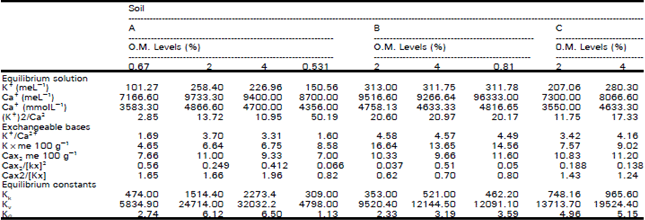
Organic Matter and Salt Concentration Effect on Exchange Selectivity Coefficient in Calcarious Soils
Department of Chemistry, Department of Soil Science Uuniversity of Agriculture, Faisalabad, Pakistan
H. Nawaz
Department of Chemistry, Department of Soil Science Uuniversity of Agriculture, Faisalabad, Pakistan
G. Murtza
Department of Chemistry, Department of Soil Science Uuniversity of Agriculture, Faisalabad, Pakistan
K. M. Zia
Department of Chemistry, Department of Soil Science Uuniversity of Agriculture, Faisalabad, Pakistan