Research Article
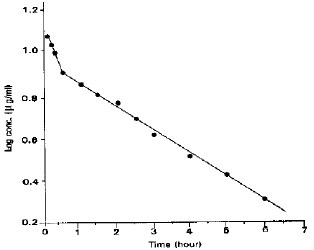
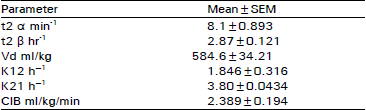
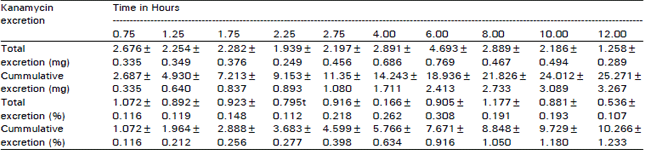
Studies on Biodisposition Kinetics and Urinary Excretion of Kanamycin in Goats
Department of Biochemistry, University of Agriculture, Faisalabad-38040, Pakistan
Tahira Iqbal
Department of Biochemistry, University of Agriculture, Faisalabad-38040, Pakistan
Munir A. Shiekh
Departrnentuf Physiology and Pharmacology, University of Agriculture, Faisalabad-38040, Pakistan
M. Nawaz
Department of Biochemistry, University of Agriculture, Faisalabad-38040, Pakistan
Amer Jamil
Department of Biochemistry, University of Agriculture, Faisalabad-38040, Pakistan