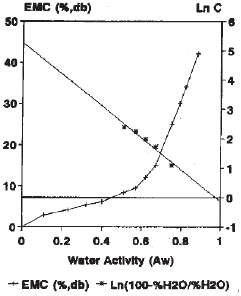
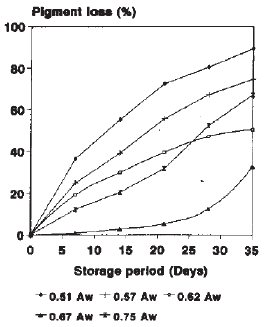
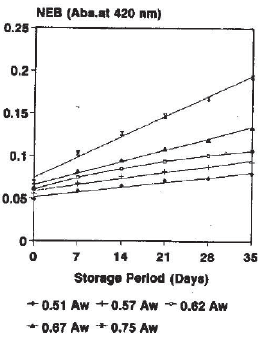
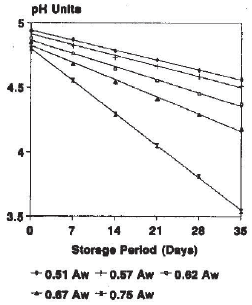
Research Article
Stability of Tomato Powder at Intermediate Moisture Levels
Department of Food Chemistry and Environmental Sciences, University of Glasgow, Scotland, UK
Musa Kaleem Baloch
Department of Chemistry, Gomal University, D.I.Khan, Pakistan
Shahzada A. Saleem
Food Technology Section, Agricultural Research Institute, D.I.Khan Pakistan
Ahmad K. Baloch
Department of Food Science & Technology, Gomal University, D.I.Khan, Pakistan