Research Article
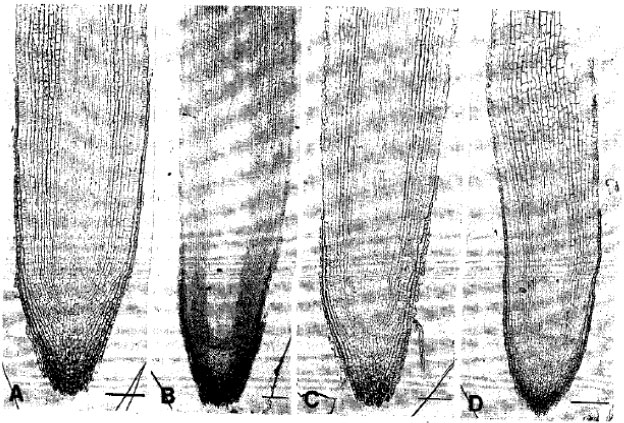
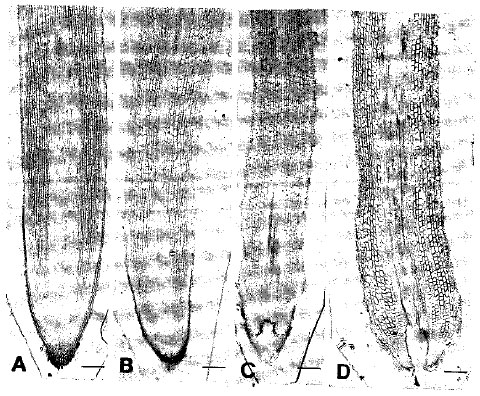
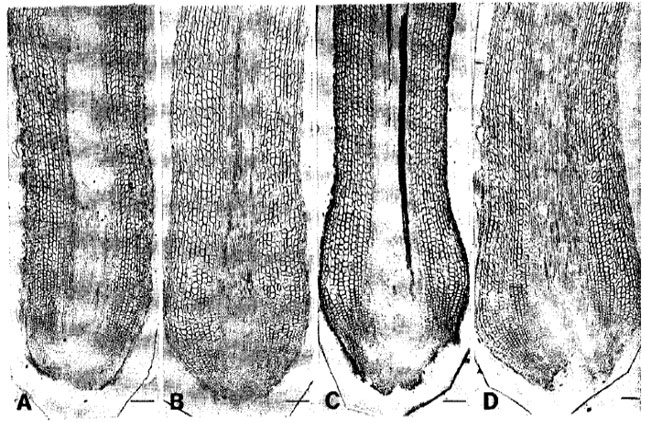
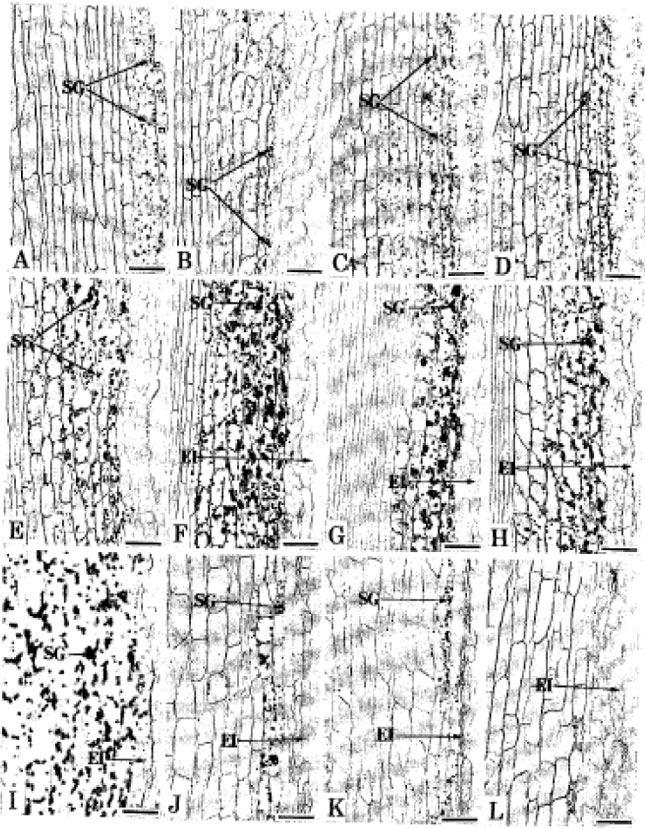
pH Optima for Citrus Inferred from the Anatomical Records of Young Roots of Trifoliate Orange Budded with Satsuma Mandarin
The United Graduate School of Agricultural Sciences, *Faculty of Agriculture, Ehime University, 3-5-7 Tarumi, Matsuyama 790-8566, Japan
Masaya Shiraishi
The United Graduate School of Agricultural Sciences, *Faculty of Agriculture, Ehime University, 3-5-7 Tarumi, Matsuyama 790-8566, Japan