Research Article
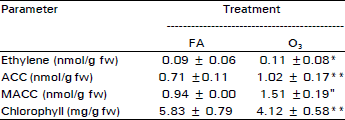
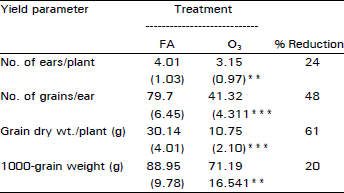
Effect of O3 on Ethylene Biosynthesis and Yield of Egyptian Cultivar of Wheat (Triticum aestivum L.)
Department of Botany, Alexandria University, 21526 El-shatby, Alexandria, Egypt
S. Anttonen
Department of Ecology and Environmental Science, Univeristy of Kuopio, P.O. Box 1627, Fin-70211, Kuopio, Finland.
M.R. Ashmore
Imperial College Centre for Environmental Technology, Th Huxley School of Environment, Earth Science and Engineering, 48 Prince`s Garddens, London Sw7 2 Pe. Uk.
J.N.B. Bell
Imperial College Centre for Environmental Technology, Th Huxley School of Environment, Earth Science and Engineering, 48 Prince`s Garddens, London Sw7 2 Pe. Uk.
J. Bender
Department of Ecology and Environmental Science, Univeristy of Kuopio, P.O. Box 1627, Fin-70211, Kuopio, Finland.
H.J. Weigel
Institute of Ecotoxicology, Fal, Bundesailee 50, D-38116, Braunschweig, Germany