Research Article
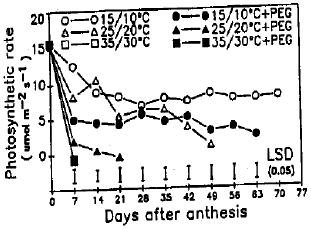
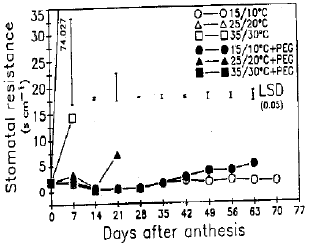
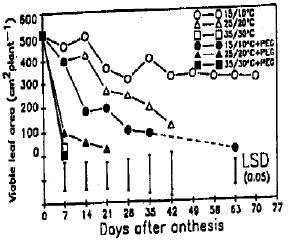
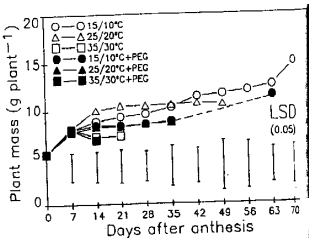
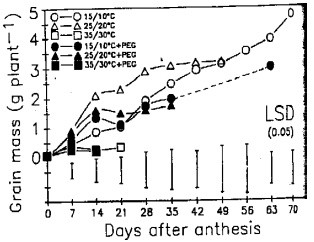
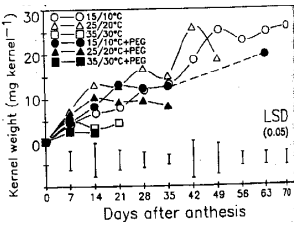
Response of Wheat to Combined High Temperature and Osmotic Stresses During Maturation. 1: Plant Photosynthesis and Productivity
Agricultural Research Institute, D.I.Khan, Pakistan
G.M. Paulsen
Department of Agronomy, Kansas State University, Manhattan, Kansas, 66506, USA