Research Article
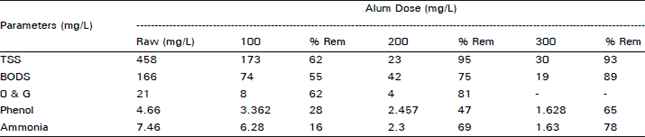
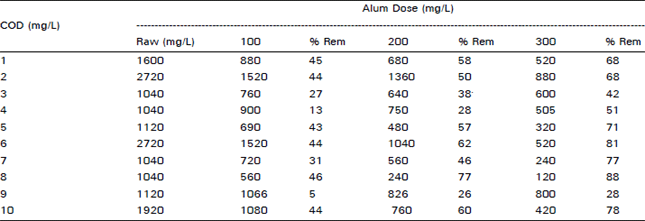
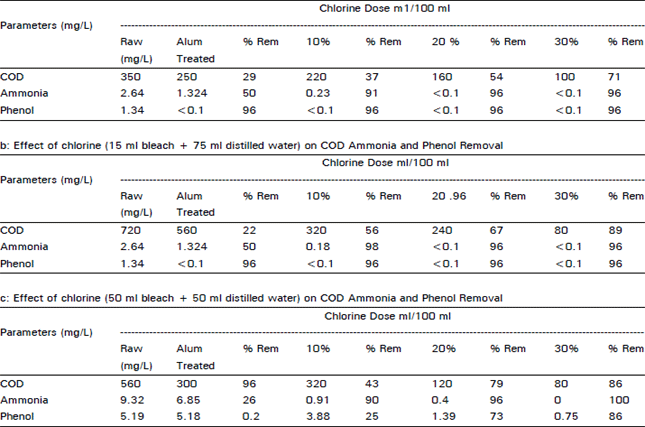
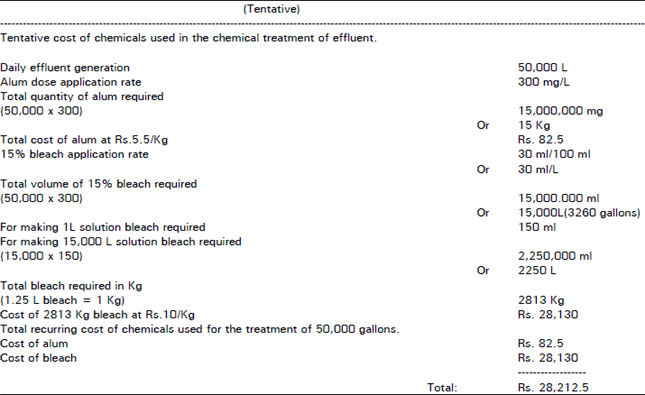
Chemical processing of Pharmaceutical wastewater for Pollution control
Institute of Environmental Studies, University of Karachi, Karachi-75270,
S. Nazrul Hasnain
Institute of Environmental Studies, University of Karachi, Karachi-75270,
Moazzam Ali Khan
Department of Biochemistry, University of Karachi, Karachi-75270, Pakistan.
M. Altaf Khan
Institute of Environmental Studies, University of Karachi, Karachi-75270,