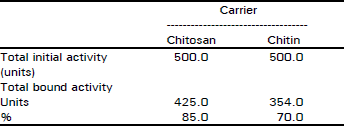
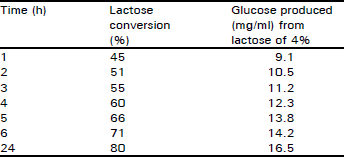
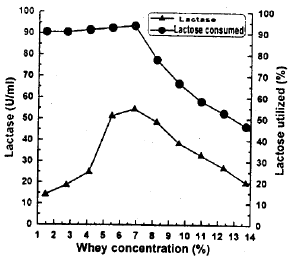
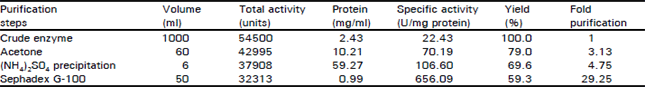Research Article
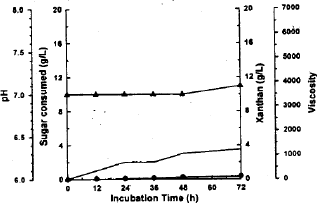
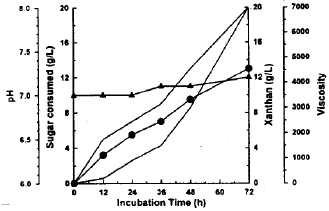
Xanthan production from whey treated with immobilized lactase
Microbiology Department, Faculty of Agriculture, Mansoura University, Mansoura, Egypt
Eman H. Ashour
Microbiology Department, Faculty of Agriculture, Mansoura University, Mansoura, Egypt