Research Article
Acoustic Ganglion In The Chick During Development and Ageing Process
Department of Anatomy, Faculty of Medicine, University of Malaya, 50603 Kuala Lumpur, Malaysia
Great many changes have been described during the development of neural structures in several animal species including primates. This includes the studies relating to the size and density of neural elements (Cowan, 1973), embryonic origin (Cowan and Wenger, 1967), different histogenic characteristics (Gaik and Farbman, 1973), distribution of cytoplasmic organelles (Hamburger, 1958) and neuronal death (Hamburger, 1961). However, most workers have confined their studies to certain selected stages of development or postnatal growth. The present study is aimed at analysing the structural organization occurring in the acoustic ganglion of the chick during embryonic development through adulthood so as to formulate the significance of these changes. It is expected that this study would form a basis on which further investigation on its applied aspects will be followed.
The chicks Gallus gallus domesticus, White Leghorn breed were used in this study. Fertilised eggs were incubated. After every 24 hours, it was considered as Embryonic Day 1 (E1), Embryonic Day 2 (E2) etc till hatching (H). Embryos till hatching were removed carefully and fixed in 10 percent formaldehyde solution at least for two weeks. Larger (older) embryos were cut transversely into suitable smaller pieces and labeled serially for future orientation. The tissues of older embryos (i.e., E15 and onwards till adult) were usually decalcified after fixation. Serial sections of 8-10 microns were stained by Cresyl Fast Violet for Nissl granules. Only a few selected stages that showed some remarkable changes are described in this work. These include E6, E8, E10, E13, E15, E18, chicks, on the day of hatching (H) and adult (A). In all, three animals in each group, having a total of twenty-four animals were used. Ganglia of both sides in each animal were used for observation. Every section of the ganglion was observed and drawn. The cells are plotted in a diagram with the help of a light microscope having a camera lucida attachment. Different categories of neurones were classified into dark and light neurones according to the difference in the intensity of cytoplasmic stain. Each of these types is again subdivided into various subclasses represented in the diagram by a symbol. Only those cells having a clear nucleus and a nucleolus were counted and measured with the help of an eye-piece graticule. The dimension of every cell was determined by calculating the average of the two measurements: one taken on its long axis at the place where the nucleus and its nucleolus were very clearly observed and another taken at an angle perpendicular to this long axis. However, the possible error in calculating the size of the cells is considered to be very minimal or negligible. The following categories of cells were classified. Tiny (<5 microns), very small (6-10 microns), small (11-15 microns), medium sized (16-20 microns), big (21-25 microns), very big (26-30 microns). The categorisation of cells on the basis of size with a uniform difference of 5 microns was initially maintained just for the sake of convenience. However, this proved to be very useful in that, the behavior of cells, especially that of the very-small cells, is very interesting on the day of hatching (uniformly) in all the ganglia studied. This explains that this particular stage of cellular growth (very-small cell stage) is a critical period during development, indicating a stage of active cell-process-formation (axon-formation) beginning to establish functional connections with the target tissues. This information is also used to interpret the functional significance of the occurrence of light cells during early stages of development.
Results
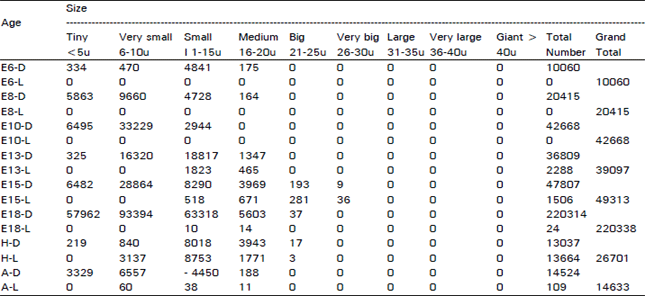
Population density (P. D = No. of cells/volume) for certain types of cells is given in parenthesis. When the dark neurones alone are present in the ganglion, they are represented just by their numbers; when they are mixed with light neurones, D = dark neurones and L = light neurones (Table 1). The ganglion showed great difference in different age groups of animals and in different areas in the same ganglion. The most striking changes are as follows. The acoustic ganglion could be clearly recognized on E6 while it had a rostro-caudal length of 0.312 mm and a volume of 0.0085 mm3. The ganglion had 10060 cells (P. D = 1183529) and all of them were dark type. In all, there were 334 (3.32%) tiny cells (P. D= 39294), 4710 (46.82 %) very small ones (P. D = 554118), 4841 (48.12 %) small ones (greatest P. D= 569529) and 175 (1.74 %) medium sized ones (P. D = 20588). On E8, the ganglion had a length of 0.416 mm, a volume of 0.0165 mm3 and 20415 cells (P. D= 1237273) and all the cells were dark type. In all, there were 5863 (28.72%) tiny cells (P. D =355333), 9660 (47.32%) very small ones (greatest P. D=585455), 4728 (23.16%) small ones (P. D= 286545) and 164 (0.8%) medium sized ones (P. D -9939). On E10, the ganglion had a length of 0.558 mm, a volume of 0.0287 mm3 and 42668 cells (P. D = 1486690) and all of these cells were dark type. In all, there were 6495 (15.22%) tiny cells (P. D = 226307), 33229 (77.88%) very small type (greatest P. D = 1157805) and 2944 (6.9%) small ones (P. D = 102578). On E13, the ganglion had a length of 0.600 mm, a volume of 0.0537 mm3 and 39097 cells (P. D= 728063). Among these cells, 36809 (94.15%) were dark type (P. D = 685456) and 2288 (5.85%) were light ones (P. R42607). In all, there were 325 (0.83%) tiny cells (P. D = 6052), 16320 (41.74%) very small type (P. D = 303911), 20640 (D= 18817 + L= 1823) (52.79%) small ones (greatest P.D = 384358) and 1812 (ID= 1347 + L= 465) (4.63%) medium sized ones (P. D = 33743). On E15, the ganglion had a length of 0.700 mm, a volume of 0.0769 mm3 and 49313 cells (P. D = 641261). Among these cells, 47807 (96.95%) were dark type (P. D = 621677) and 1506 (3.05%) were light ones (P. D= 19584). In all, there were 6482 (13.14%) tiny cells (P. D = 84291), 28864 (58.53%) very small ones (greatest P. D = 375345), 8808 (D = 8290+ L = 518) (17.86%) small ones (P. D= 114538), 4640 (D = 3969 + L = 671) (9.41%) medium sized ones (P. D= 60338), 474 (P.D = 193+L= 281) (0.96%) big ones (P. D= 6164) and 45 (D= 9 + L = 36) (0.09%)) very big ones (P. D= 585). On E18, the ganglion had a length of 0.720 mm, a volume of 0.2386 mm3 and 220338 cells (P. D= 923462). Among these cells, 220314 (99.99%) were dark type (P. D = 923361) and 24 (0.01%) were light ones (P. ID = 101). In all, there were 57962 (26.31%) tiny cells (P. D= 242925), 93394 (42.39%) very small ones (greatest P. D = 391425), 63328 (D = 63318 + L = 10) (28.74 %) small ones (P. D= 265415), 5617 (D = 5603 + L = 14) (2.55%) medium sized ones (P. D= 23542) and 37 (0.02%) big (dark) ones (P. D = 155). On the day of hatching, the ganglion had a length of 0.910 mm, a volume of 0.1568 mm3 and 26701 cells (P. D = 170287). Among these cells, 13037 (48.83%) were dark type (P. D= 83144) and 13664 (51.17%) were light ones (P. D= 87143). In all, there were 219 (0.82%) tiny cells (P. D = 1397), 3977 (D = 840 + L = 3137) (14.89%) very small ones (P. D= 25363), 16771 (D = 8018 + L = 8753), (62.81%) small ones (greatest P. D = 106958), 5714 (D= 3943 + L = 1771) (21.4 %) medium sized ones (P. D = 36442) and 20 (D = 17 + L = 3) (0.07%) big ones (P. D = 127). In the adult situation, the ganglion had a length of 1.000- mm, a volume of 0.1420 mm3 and 14633 cells (P. D = 103050). Among these cells, 14524 (99.26%) were dark type (P. D = 102282) and 109 (0.74%) were light ones (P. D = 768). In all, there were 3329 (22.76%) tiny cells (P. D = 23444), 6617 (D = 6557 + L+ 60) (45.22%) very small ones (greatest P. ID = 46599), 4488 (D= 4450 + L= 38) (30.67%) small ones (P. D= 31606) and 199 (D= 188 + L = 11) (1.36%) medium sized ones (P. D = 1401).
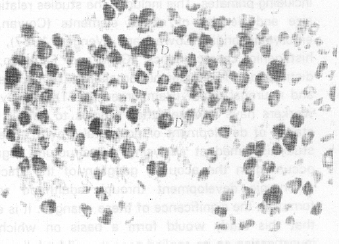 | |
| Fig. 1: | Shows Dark (D) and Light (L) cells observed in the Ganglion. Different size-categories of cells are also seen. Each division of the scale-bar is equal to 10 microns |
From the behaviour of the dark and light cells in the acoustic ganglion during development and growth, it is presumed that the dark cells represent a group of active cells and the light cells represent a group of inactive, dying, dead or degenerating cells. The ganglionic volume, its rostro-caudal length, cross-sectional dimensions and cellular constitution vary greatly from time to time at different stages of development.
| Table 1: | Illustrates the total number of dark and light cells in the acoustic ganglion in different age groups of animals in the ontogeny of the chick. (D = Dark Cells, L = Light Cells, E = Embryonic Age, H= Day of Hatching, A = Adult) |
 | |
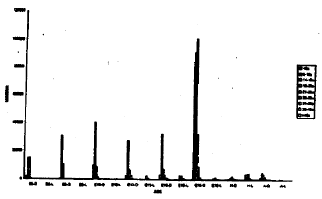 | |
| Fig. 2: | Total number of dark and light cells in the acoustic ganglion in different age groups of animals in the ontogeny of the chick |
All the cells observed on E6 through E10 are dark type and no light cells are represented among them (Fig. 1,2).The light cells appear for the first time, on E13, and continue to be present afterwards through the whole life afterwards. The dark and light cells are disposed at random throughout the ganglion as from E13 onwards. However, some investigators (Gaik and Farbman, 1973) have found such random distribution only in mature (from 18th day of incubation to adult) ganglion, i.e., from shortly before hatching to adult and not in the younger stages.
The tiny cells are found to be always dark. The very-small type of cell is also found to be dark through the whole embryonic period till E18. Later, on the day of hatching, the light cells have appeared among this very-small type of cells as well and these light cells continue to be present even in the adult situation. This suggests that the very-small type of cells appear to keep them to be an active group till the day of hatching and be ready to replace the inactive or dead cells. However, degeneration begins among this group also as from the day of hatching. Normally, there cannot be any necessity for the establishment of new functional projections after the day of hatching, since all these connections might have been completely established by this time while the animal is prepared and ready to lead an independent living. Therefore, there is no need for further growth and maturation of these smaller categories of cells, and therefore the cell death begins (this being represented by tke occurrence of light cells) even among these very-small cells to prevent further growth (that seems unnecessary).
On E8, the larger classes of cells have reduced in number. This might suggest that the cells that fail to form functional connections might be immediately removed from the ganglion. That is, this loss of cells might be due to their failure to form functional connections. It appears that the stages between E8 and E10 are critical periods for the establishment of proper functional connections and therefore, the cell death and phagocytosis are also similarly so rapid. It may be recalled here that in the trigeminal ganglion, by the 8th day of incubation at least some of the ganglion cells have established peripheral and central connections as evidenced by the presence of reflexogenic responses to tactile stimulus of the beak (Hamburger and Narayanan, 1969). Similarly a few authors (Noden, 1980) have found that many trigeminal sensory cells have extensive peripheral projections by the end of the first week of incubation and suggests that these cells must cease dividing very early in embryonic development. It is during this period (E8) that the light cells have appeared in the trigeminal ganglion for the first time in the present series of investigation (Pillay, 1999) as well. Coincidence of the occurrence of light cells in the trigeminal ganglion for the first time on E8 during development (Pillay, 1999) and the first appearance of reflexogenic response indicating the establishment of peripheral connections (Hamburger and Narayanan, 1969; Noden, 1980) during the same developmental stage (E8) might clearly suggest that the cells that fail to establish functional connections might become inactive, lose their functions, die, finally degenerate and disappear from the ganglion and, that the so-called light-cells represent the beginning stage of cell degeneration.
The medium sized class of cells is one of the critical stages in the development of the ganglion while probably an attempt to establish proper connections with the projection-fields is taking place. However, when this attempt fails, these cells are unable to grow further nor live longer and so become inactive and die. This resembles the suggestion of a few investigators (Hamburger and Levi-Montalcini, 1949; Cowan, 1973; Cowan and Wenger, 1967) that cell degeneration is probably influenced by the peripheral fields of innervation and that the cell death can be influenced by the target cells of a neurone population.
Even though during E10 there is an increased cellular death as indicated by the great loss of small as well as medium sized cells, the loss is compensated by the increased production and growth of tiny cells so that the total number of cells finally shows an increase. This is somewhat similar to the suggestion of a few investigators (Rubel et al., 1976) about cell death which they observed in the brain-stem auditory nuclei where most cell-death occur in the period between E 10 - E13.
The volume shows gradual increase up to E18 while the ganglion reaches its maximum size. Later, on the day of hatching, the volume has dropped down (because of reduced average cross sectional diameter). In the adult situation also, the volume has again a set-back (having a reduced cross-sectional diameter). Thus the volume increases through the whole embryonic period whereas it drops down on the day of hatching and continues to reduce through post-hatching period till adulthood. This condition is very peculiar to the acoustic ganglion. This is very different from that observed in other cranial nerve ganglia where the ganglion is the largest in the adult situation (Pillay, 1999). This is possibly due to the relatively much reduced activity of the acoustic function due to ageing process. The ganglion-size increase is minimal during the beginning of development, i.e., up to E10, while as from E13 onwards, the volume shows a quicker and greater increase. This suggests that the increase in the ganglionic volume during this period is basically due to the increased size of the neuropil (which includes cell-processes and neuroglia) and the increased size of the cell-body.
It is also evident from the results of some investigators (Hamburger and Levi-Montalcini, 1949; Rubel et al., 1976) that major changes in cell-size, nuclear size and cell-number occur between E13 and the day of hatching in different cell-groups (nuclei) in the central nervous system. The "nuclear size" of these investigators might be correlated with the "ganglion-size" in the present study because corresponding and parallel changes are taking place in the brain-stem auditory nuclei and the peripheral ganglionic projection in the chicken (Rubel et al., 1976). The present results in the acoustic ganglion also show similar tendency. The ganglionic volume increases greatly when the cell-death comes to an end around E13 so as to reach its maximum size (0.2386 mm3) on E18. This observation is similar to that described by a few earlier investigators (Rubel et al., 1976) in the brain-stem auditory nuclei in the chicken. Similar synchronous growth of the functionally related cell-groups has also been described in visual system of golden hamster (Ptacek and Fagan-Duban, 1974) where cell-density decreases and, cell-size increases in both the visual cortex and superior colliculus in a parallel fashion.
When the attempt to establish functional connection fails, these cells are unable to grow further and become inactive and die. The cellular death and degeneration might begin even during the early stages of development (on E8 or earlier) as a result of their failure to establish functional projection. And therefore, it appears that the period of accelerated degeneration of cells is also the period of active establishment of functional connections. Stages between E10 - E13 are probably active periods of establishment of functional connections. The growing neurones or larger classes of neurones might represent those cells that have succeeded in establishing functional connections.
There is a tremendous increase in the total number of cells' on E18 and that this increase concerns mainly with the tiny, very small and small types. Main purpose of this active proliferation of smaller categories of cells on E18 is probably to produce another kind of cells with the capacity of phagocytosis in order to remove the dead cells (and unsuccessful neuronal elements that have failed to establish functional connections) so as to free the tissue from their toxic effects. Similar findings of the presence of enormous numbers of phagocytic cells at similar stages have been reported by several investigators (Chu-Wang and Oppenheim, 1978; O'Connor and Wyttenbach, 1974).
There is a huge loss of cells on the day of hatching. This loss mainly concerns with those cells which have made their appearance on E18, that is, those tiny, very small and small cells, most of which might represent phagocytic cells. They have an important function of removing those unsuccessful neuronal elements while the animal is prepared for an independent living. Once their function of removing those unwanted or unsuccessful neuronal elements is completed, they themselves disappear, probably by self-destruction, i.e., they themselves undergo degeneration and disappear from the tissue. The cellular death (and degeneration) could be one of the factors that regulate the size of cell population (Hamburger and Levi-Montalcini, 1949) and is influenced by the peripheral field of innervation and thus has a role in morphogenesis.
In many situations through the whole ontogeny of the chick it is observed that new cells are being formed or added by active proliferation of tiny cells with some interrupted periods of rest or reduced proliferation while the total number of cells is actually not declining. This clearly supports the view of some investigators (Hamburger, 1958) that there are degenerating cells at a time when the number of cells was not actually declining and that new cells must have been entering, Similarly in Xenopus, the number of degenerating cells was greatly in excess of that required to account for a decline in cell number and therefore, concluded that the turn-over was taking place in the developing ventral horn (Hughes, 1961). It is noticed that there are more than one period of active proliferation and active degeneration which are indicated by the sharp fluctuation (rise and decline) in the number of cells in different stages during development and growth. It is also strange to note that the very big type of cells is never observed again either on the day of hatching or in the adult situation explaining that these cells are a group of unnecessary aberrant formations, not essential for the functioning of acoustic ganglion.
In the adult situation, there is a great reduction in the total number of cells and also among medium sized (and big) cells. The big cells have been totally removed from the adult ganglion, probably as a result of ageing process. Possibly the larger classes of cells are not functionally active and so not important in the acoustic ganglion. Therefore, the main target of elimination is on these larger categories of cells during ageing process.
The tiny and very small types of cells have increased in the adult situation in spite of the reduction in the total number of cells. The presence of increased number of tiny and very small type of cells in the adult ganglion possibly indicates that many primitive cells might be continuously kept alert and reserved in order to replace those cells which lose their functional activity due to ageing process. This is possibly because of the functional importance of this ganglion for the normal behaviour of the animal. It appears that there is a continuous attempt to replace the older cells by newly formed ones. This is a striking difference from the constitution and behaviour of other cranial nerve ganglia. This suggests that the acoustic neurones are more sensitive to damage by ageing process than those in other ganglia.
The author wishes to express his sincere and unlimited gratitude to Professor. Dr. Dubbeldam JL, Leiden University, Netherlands for his continued generosity, guidance, cooperation and support throughout this study. His continued efforts and encouragement to make it as part of a thesis for the award of a DSc degree of Leiden University are greatly appreciated.