ABSTRACT
Growth rate and branching of D. uninucleata were correlated to the exocellular concentration of calcium in culture medium. Calcium concentrations of 1-10 mM stimulated germ tube formation and reduced the lag phase. Mycelia grew in media amended with 1-0 mM Ca2+ were highly branched and the number of cells/hypha together with the number of cells/branch significantly increased. Gametangial formation have been induced by 10-40 mM Ca2+ and the highest percent of ascus formation with the longest asci was achieved in the presence of 10 mM calcium. Increasing Ca2+ ions concentration above 40 mM had no further effect on growth aspects yet concentrations higher than 160 mM were toxic and partially inhibited growth, branching and reproduction.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.1999.1159.1163
URL: https://scialert.net/abstract/?doi=pjbs.1999.1159.1163
INTRODUCTION
The role of calcium ions in the metabolism of living organisms is in diverse (Bygrave, 1978), but it is increasingly apparent that Ca2+ is important in control at the cellular level (Fletcher, 1979; Holmes and Stewart, 1979). The response to Ca2+ depend on the relative capacities of the cells to accumulate calcium and the initial adsorption to the cell surface is related to the number of available binding sites at the cell wall which may be different even in the strains of the same species (Pitt et al., 1988). Several reports suggest the involvement of Ca2+ in maintaining polarized hyphal growth (Schmid and Harold, 1988; Jackson and Heath, 1989) and its importance, for growth, metabolism and differentiation (Saavedra-Molina et al., 1983; Griffith et al., 1988; Pitt and Kaile, 1989; Masy et al., 1991).
Favre and Turian (1987) reported that Ca2+ has a regulatory role in the fungus Neurospora Crassa. The addition of Ca2+ ions to submerged cultures, induced spore germination in penici/lium notatum (Gilbert and Hickey, 1946) and initiated spore production in Penicillium cyclopium (Schroeder, 1978). Davis et al. (1986) suggest that Ca2+ is an important regulatory ion in yeast. It was also reported that external level of Ca2+ induced branching in Neurospora and Achlya and this response was due to the release of Ca2+ from an intracellular reservoir (Reissig and Kinney, 1983). Dipodascopsis uninucleata is an intermediate between single celled system of the yeast and the complex mycelium system of the euascomycetes (Biggs, 1937). Ascopores of D. uninucleata germinate to give short hyphae along which lateral branches and gametangia are initiated in a certain pattern (Elwy, 1980). Sometimes the pair of gametangia fail to unite or only single gametangium is formed and these elongate to form lateral branches.
The fungus was used to study the effect of Ca2+ on the hyphal extension and branching in this model system.
MATERIALS AND METHODS
The fungus Dipodascopsis uninucleata Batra and Milner was obtained from C.A.B. International Mycological Institute (IMI 86676). Fresh cultures were prepared from the stock cultures and grown in MYGP liquid medium at temperature 25±1°C and 150 rpm in a rotary shaker. The defined culture medium MYGP for vegetative growth contains the following components (analar grade) in one liter of distilled water: 5 g glucose, 5 g peptone, 3 g yeast extract and 3 g malt extract.
Calcium chloride was used as the source of Ca2+ ions. The following concentrations were used: 1, 2, 5, 10, 20, 40, 80, 160 and 320 mM. Ascospores of the inoculum were obtained from 4 days-old cultures by filtration through sterile glass wool twice to remove mycelia! fragments. The density of the ascospores suspension was 106/ml. Flasks without CaCl2 were used as the control.
Measurements of growth parameters
Spore germination: Set of flasks containing 30 ml MYGP, were prepared in duplicates and amended with the different concentrations of calcium chloride, After outoclaving the flasks were inoculated with 0.2 ml of the spore suspension and incubated at 25±1°C at 150 rpm. The culture were examined microscopically at intervals. At least 300 spores were examined from each culture and the percent of germination was estimated and compared to that of the control.
Growth and reproduction: Set of flasks were prepared as described in spore germination, then incubated for 38 hr. The cultures were then examined to observe the number of cells/hypha, the number of branches/hypha and the number of cells/branch.
Another set of flasks were prepared as described in spore germination and incubated for 42 hr, then examined microscopically to record the number of successful gametangia, the number of asci and the number of failed gametangial fusion in each hypha. 60 hyphae sampled at random were examined for each treatment. The experiments were carried out 3 times and the means of the readings were calculated.
Colourimetric estimation of growth biomass: In this study triplicate flasks were prepared for each treatment as described in the study of spore germination. The flasks were incubated for 50 h then the produced biomass was measured using spectrophotometer (spectronic 20) at the wavelength 500 nm. The experiment was carried out 3 times and the means of the readings were calculated.
Statistical analysis: All data are given as the mean±standard deviation and the difference between the means were tested using the t test at 95 percent confidence limit.
RESULTS
The results of Table 1, show that Ca2+ in concentrations upto 80 mM decreased the lag period of ascospores and stimulated the process of ascospores germination. Ca2+ levels up to 40 mM active significantly germination and 100 per cent of the ascospores were able to germinate in 18 h incubation compared to 89 percent for the control. 80 mM Ca2+ level was also stimulatory during the first 12 h of incubation but longer periods showed non-significant differences with the control. As the concentration increased to 160 mM and higher, a clear inhibitory effect was observed especially when Ca2+ level reached 320 mM, at which a delay in the onset of germination was clear and only 40 percent of the spores were able to germinate compared to 89 percent of the control.
Table 2 showed the response of fungal growth to various concentrations of Ca2+ ions. considering the number of cells/hypha, the most effective concentration was 10mM, it produced significant increase in the number of cells/hypha in comparison to the control. Higher concentrations of Ca2+ up to 40 mM were also stimulatory. However Ca2+ levels of 160 and 320 mM produced marked decrease in cell number/hypha.
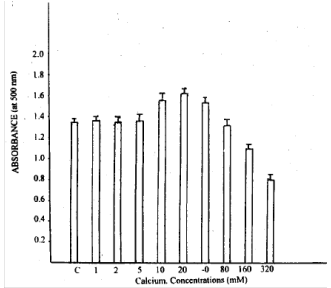 | |
| Fig. 1: | Growth biomass of D. uninucleata at different calcium concentration. Arrows indicate standard deviation |
The percent of branched hyphae in cultures amended with Ca2+ up to 80 mM was 100 percent while that of the control was 92 percent. On the other hand, 160 and 320 mM Ca2+ inhibited branch initiation by 12 and 20 percent respectively. The table showed also the number of branches along each hypha. A remarkable increase in the number of branches was observed in the presence of Ca2+ at a level between 10-40 mM. Higher concentrations (160 - 320 mM) resulted in a decrease in the number of branches (laterals). The effect of Ca2+ on the number of cells/branch was determined and Ca2+ at 10 mM concentration increased the average length of laterals by one cell.
The results of Table 3 illustrates the effect of various Ca2+ concentration on sexual reproduction of D. uninucleata. Ca2+ ions induced the formation of gametangia, so it increases the number of attempts especially at levels 10-40 mM. However, higher concentrations reduced the number of attempts, in other words, retarded sexual reproduction. Also, number of successful fusion or failure attempts/hyphae were investigated. Number of successful attempts increased markedly at levels 10, 20, 40 mM. On the contrary, reduction in the failed attempts was observed with the concentrations 160 and 320 mM. The highest number of asci/hypha was observed at cultures with 10 mM also 20 and 40 mM increased the number of asci significantly.
Fig. 1 shows the growth represented by the absorbance at 500 nm, it is clear that low concentrations of calcium (1-5 mM) had no effect on growth, while, 10-40 mM markedly increased the growth. High levels of Ca2+ decreased the growth of the fungus. Figure 2 illustrates the effect of Ca2+ at concentrations 10m, 80 and 320 mM on growth, branching and ascus formation in cultures incubated for 48 h and compared with the control of the same age. The longest asci developed in the presence of 10 mM Ca2+ while 320 mM Ca2+ delayed ascus formation.
DISCUSSION
Fungi appear not to have an absolute requirement of Ca2+ for growth and a low internal concentration is sufficient for calcium dependent events Ascospores of D. uninucleata can grown in media containing no calcium but this does not mean that they do not need Ca2+ as it may be present in growth media as contamination from other chemical constituents. The present investigation showed that exogenous Ca2+ ion stimulated spore gemination, hyphal extension, branching and sexual reproduction in D. uninucleata, similar results were reported for other fungi (Pitt and Ugalde, 1984; Schmid and Harold, 1988). concentrations from 1-10 mM Ca2+ increased the rate of spore germination and produced synchronus cultures. This confirm the report of Gadd and Brunton (1992) who found that 5 mM Ca2+ stimulated spore germination in Ophiostoma ulmi. Calcium also induced germ tube formation even in non germination medium of Ceratocystis ulmi (Muthukumar and Nickerson, 1984).
Hyphal extension and lateral branching of D. uninucleata seem to need higher Ca2+ concentration than spore germination (10-40 mM), Similar stimulation of hyphal extensions in Neurospora crassa and saprolegnia ferax was observed by Schmid and and Harold (1988) and Jackson and Heath (1989). Also high branching in Fusarium graminearum was obtained using 1.4 x 10–8 M Ca2+.
Fungal hyphae grow in two modes, linear extension and branching. The precursor for cell wall synthesis and plasma membrane are packaged in vesicles and transported to the extreme tip where vesicle fusion and exocytosis of their contents take place for hyphal extension, but lateral branching is complicated and little is known about this process in fungi (Trinci, 1978; Gooday, 1983).
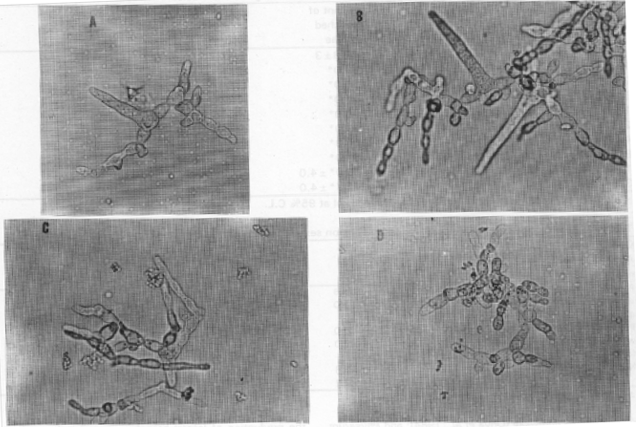 | ||||
| Fig. 2: | Light-micrographs of hyphae and Asci with Ca2+ after 48 hrs | |||
| a) untreated | b) 10 mM Ca+2 x 2000 | c) 80 mM C2+ | d) 320 mM Ca+2 | |
| Table 1: | The effect of different concentrations of calcium on spore germination of D. uninucleata |
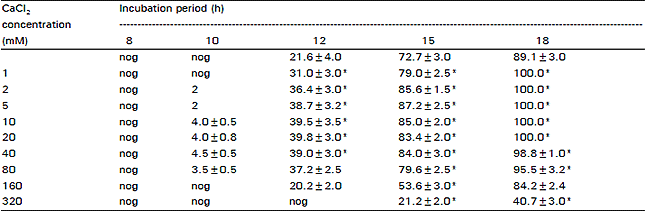 | |
| St.D nog = No germiration *Significant difference with the control at 95 % C.L. | |
A mechanism by which Ca2+ affects extension had been suggested by Robson et al. (1991). It could be the result of an interaction with the cytoskeleton at the apex, an effect on vesicle distribution, or on wall assembly. Calmodulin which is a calcium binding protein was detected in many yeast and filamentous fungi (Hubbard et al., 1982) and proved to have a role in germination and growth of Manisopliae (St Leger et al., 1989). Calmodulin is also important in regulating morphogenesis of Candida albicans (Paranjape et al., 1990) as well as other fungi (Wessels, 1986).
Ca2+ calmodulin interaction play an important role in nuclear division, bud enlargement and differentiation in Saccharmoces cerevisae (Ohya et al., 1984) and Physarum polycephalum (Uyeda and Furuya, 1986).
| Table 2: | The effect of different concentrations of calcium on branching of D. uninucleata |
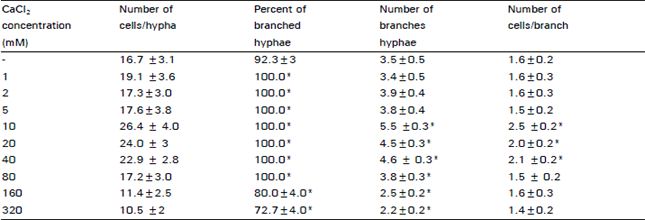 | |
| ± St.D *Significant difference with the control at 95% C.L. | |
| Table 3: | The effect of different calcium concentrations on sexual reproductio in D. uninucleata |
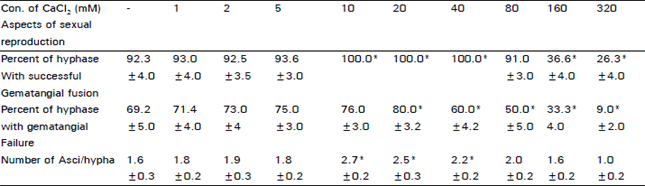 | |
| ± St.D*Significant difference at 95% C.L. | |
Stimulation of sexual reproduction in D. unincleata was observed by calcium concentrations rang from 10 to 40 mM, similar observations was reported for penicillium notatum (Pitt and Poole, 1981). They reported that 10 mM Ca2+ induced synchronous phialide and conidium development. Ca2+ also plays a role in the production of phialides and microconidia in F. graminearum (Robson et al., 1991).
Increasing Ca2+ concentration above 160 mM decreased germination, branching and differentiation in D. uninucleata which indicates its toxic effect at high concentrations. Yoh-Ichi and lsao (1990) reported that growth of S. cerevisae mutant was suppressed by high concentrations of calcium. also, toxicity may be caused by concentration (>40 mM) as observed by Pitt et al. (1988) in the case of P. notatum. This inhibitory effect of high concentrations of extracellular Ca2+ may be due to its effect on the sensitive transport of wall vesicles or indirectly by an action n the actin cytoskeleton (Jackson and Heath, 1989).
In conclusion Ca2+ at 10 mM level was stimulatory to all morphogenesis processes and this may be a response to an intracellular signal which triggers the hyphal tip extension, the production of lateral branches, gametangia, asci and ascospores. Further investigations will be needed to detect the presence of calmodulin in the cells and its interaction with Ca2+ in producing regulatory effect on morphogenesis of D. uninucleata.
REFERENCES
- Bygrave, F.L., 1978. Mitochondria and the control of intracellular calcium. Biol. Rev., 53: 43-79.
CrossRefDirect Link - Masy, C.L., M. Kockerols and M.M. Mestdagh, 1991. Calcium activity versus calcium threshold as the key factor in the induction of yeast flocculation in simulated industrial fermentations. Can. J. Microbiol., 37: 295-303.
CrossRefDirect Link - Davis, T.N., M.S. Urdea, F.R. Masiarz and J. Thorner, 1986. Isolation of the yeast calmodulin gene: Calmodulin is an essential protein. Cell, 47: 423-431.
CrossRefDirect Link - Favre, B. and G. Turian, 1987. Identification of a calcium-and phospholipid-dependent protein kinase (protein kinase C) in Neurospora crassa. Plant Sci., 49: 15-21.
CrossRefDirect Link - Fletcher, J., 1979. An ultrastructural investigation into the role of calcium in oosphere-initial development in Saprolegnia diclina. Microbiology, 113: 315-326.
CrossRefDirect Link - Gadd, G.M. and A.H. Brunton, 1992. Calcium involvement in dimorphism of Ophiostoma ulmi, the Dutch elm disease fungus and characterization of calcium uptake by yeast cells and germ tubes. Microbiology, 138: 1561-1571.
CrossRefDirect Link - Gilbert, W.J. and R.J. Hickey, 1946. Production of conidia in submerged cultures of Penicillium notatum. J. Bacteriol., 51: 731-733.
Direct Link - Griffith, J.M., J.R. Iser and B.R. Grant, 1988. Calcium control of differentiation in Phytophthora palmivora. Arch. Microbiol., 149: 565-571.
CrossRefDirect Link - Holmes, R.P. and P.R. Stewart, 1979. The response of Physarum polycephalum to extracellular Ca2+: Studies on Ca2+ nutrition, Ca2+ fluxes and Ca2+ compartmentation. Microbiology, 113: 272-285.
CrossRefDirect Link - Hubbard, M., M. Bradley, P. Sullivan, M. Shepherd and I. Forrester, 1982. Evidence for the occurrence of calmodulin in the yeasts Candidas albicans and Saccharomyces cerevisiae. FEBS Lett., 137: 85-88.
CrossRefDirect Link - Jackson, S.L. and I.B. Heath, 1989. Effects of exogenous calcium ions on tip growth, intracellular Ca2+ concentration and actin arrays in hyphae of the fungus Saprolegnia ferax. Exp. Mycol., 13: 1-12.
CrossRefDirect Link - Muthukumar, G. and K.W. Nickerson, 1984. Ca (II)-calmodulin regulation of fungal dimorphism in Ceratocystis ulmi. J. Bacteriol., 159: 390-392.
Direct Link - Ohya, Y., Y. Ohsumi and Y. Anraku, 1984. Genetic study of the role of calcium ions in the cell division cycle of Saccharomyces cerevisiae: A calcium-dependent mutant and its trifluoperazine-dependent pseudorevertants. Mol. General Genet. MGG., 193: 389-394.
CrossRefDirect Link - Paranjape, V., B.G. Roy and A. Datta, 1990. Involvement of calcium, calmodulin and protein phosphorylation in morphogenesis of Candida albicans. Microbiology, 136: 2149-2154.
CrossRefDirect Link - Pitt, D. and P.C. Poole, 1981. Calcium-induced conidiation in Penicillium notatum in submerged culture. Trans. Br. Mycol. Soc., 76: 219-230.
CrossRefDirect Link - Pitt, D., J.C. Barnes and U.O. Ugalde, 1988. Differential uptake of calcium by strains of Penicillium notatum and relationships to calcium-induced conidiation. Trans. Br. Mycol. Soc., 91: 489-499.
CrossRefDirect Link - Reissig, J.L. and S.G. Kinney, 1983. Calcium as a branching signal in Neurospora crassa. J. Bacteriol., 154: 1397-1402.
Direct Link - Robson, G.D., M.G. Wiebe and A.P. Trinci, 1991. Low calcium concentrations induce increased branching in Fusarium graminearum. Mycol. Res., 95: 561-565.
CrossRefDirect Link - Saavedra-Molina, A., R. Villalobos and M. Borbolla, 1983. Calcium uptake during the cell cycle of Saccharomyces cerevisiae. FEBS Lett., 160: 195-197.
CrossRefDirect Link - Schmid, J. and F.M. Harold, 1988. Dual roles for calcium ions in apical growth of Neurospora crassa. Microbiology, 134: 2623-2631.
CrossRefDirect Link - St Leger, R.J., D.W. Roberts and R.C. Staples, 1989. Calcium-and calmodulin-mediated protein synthesis and protein phosphorylation during germination, growth and protease production by Metarhizium anisopliae. Microbiology, 135: 2141-2154.
CrossRefDirect Link - Uyeda, T.Q. and M. Furuya, 1986. Effects of low temperature and calcium on microfilament structure in flagellates of Physarum polycephalum. Exp. Cell Res., 165: 461-472.
CrossRefDirect Link - Wessels, J.G.H., 1986. Cell wall synthesis in apical hyphal growth. Int. Rev. Cytol., 104: 37-39.
Direct Link









Mariam Farjah Reply
I would like to contact the authur of this paper, we study together for our Ph.D at the university of London back in 1980.
Since then i lost contact with her and i am looking for her
I appreciate if you have any contact information from Dr Elwy or her husband Dr Mohammd Osman