Research Article
Criteria Characterizing Tumor Cells Observed in the Helicobacter Pylori- Associated-Gastric Epithelial Cells and Predicting Tumorgenesis
Department of Zoology, Faculty of Science, Alexandria University, Alexandria, Egypt
Helicobacter pylori (H. pylori) are a spiral shaped microaerophilic gram-negative bacterium. It is often observed colonizing the human gastroduodenal mucosa (Freedberg and Barron, 1940). It is now well known that H. pylori infection is probably the most common bacterial infection in the world (Guisset et al., 1997). Association between H. pylori and gastric and duodenal mucosa was reported by numerous authors (Bartel et al., 1992, Noach et al., 1994; Janas et al., 1995).
The pathogenicity of H. pylori has recognized since 1989 in the developed world where prevalence is 20 to 40 percent (Guisset et al., 1997). Moreover, the various hypotheses of pathogenic effect of H. pylori (HP), histological and ultrastructural characteristics of changes in the gastric and duodenal mucosa in HP infection are presented (Zaitseva, 1991). The majority of 'authors considered HP as a possible pathogenic factor in chronic gastritis type B, gastric ulcer and duodenal ulcer (Guisset et al., 1997; Gasbarrini et al., 1997). They also reported that the role of organism is certain and its low recurrence rate justifies eradication.
Also, it was found that H. pylori plays an essential role in the development of neoplastic gastroduodenal pathogenesis (Tursi et al., 1997). In the last few years, moreover, besides a local tissue damage, an association between H. pylori infection and various extra intestinal pathologies has also been described (Gasbarrini et al., 1997). The diagnosis of H. pylori infection usually involves invasive (endoscopic) methods or complex procedures using carbon -14 as a radioactive test (Marshall and Surveyor, 1988). There is, therefore, a continuing interest to evaluate the fine structural changes in gastric endoscopic biopsy as a result of association with H. pylori. Transmission electron microscope was the tool used, to shed more light on the mechanism (s) of action of the organism on gastric epithelial cell ultrastructure.
Fifty patients (of both sex), aging <45 years complaining of dyspeptic manifestations or with symptoms suggesting chronic gastritis, or peptic ulcer, not receiving any drug that cause gastritis were admitted to Alexandria University Hospital for treatment. Only positive cases for H. pylori as proved by bacteriological culture and immunological test assessment were selected. Three biopsy specimens (seropositive to specific 1gG H. pylori by ELISA technique on serum samples) were obtained by upper endoscopy. Involved corresponding sites biopsy were chosen and punch biopsies under local anesthesia. For electron microscopy, small blocks of tissue were fixed in glutaraldehyde-osmium; phosphate buffered and embedded in Epon. Semithin (1 μm) sections were stained with toluidine blue, while ultrathin (50 μm) were double stained with uranyl acetate-lead citrate and investigated in a Jeol 100 CX electron microscopy.
The location and appearance of H. pylori, in gastric biopsy specimens were assessed with light and transmission electron microscope. Our preparations showed that the organisms, occurred in the lumen closely adhering to the superficial epithelial cells, of some glands (Figs. 1, 2a,b). The electron micrographs revealed that most of bacteria looked curved, approximately 0.8 microns long and 0.4 microns wide and appeared morphologically intact as double membrane bound organism, with more or less lucent protoplasmic core and dense peripheral protoplasm that contained numerous, dense granules and ribosome (Fig. 2c). Our preparations revealed also, many pathological features. The most conspicuous abnormalities were seen in the nuclei. Some cells showing segmented nuclei that lead ultimately to images suggesting fragmentation of the nucleus (Fig. 3a, b). In addition, some other nuclei with large size revealed features of chromatolysis (Fig. 3b, 4a), were euchromatin predominates. Moreover, the nuclear envelope of most cells exhibited frequent irregularities (Figs. 2a, b). In addition, one of the most predominant nuclear abnormalities was the frequent presence of nuclear pockets; such pockets, demarcated by a band of chromatin and contained cytoplasmic material. Nuclear inclusion was also visible and contained cytoplasmic structures mostly ribosomes (Fig. 5a, b). Cells with pyknotic nuclei were also observed in our preparations (Fig. 2a).
 | |
| Fig. 1: | Semithin section fixed with F4G1-and stained with toluidine blue. A portion of gastric gland of patient infected with H. pylori. . Showing gastric glands with columnar epithelial cells, the organism could be identified with difficulty, X 1000 |
As regards cytoplasmic abnormalities most cells showed a paucity of organelles; the mitochondria were few in number, pleomorphic in shape and peripherally located (Figs 3a, b). However, in other cells both mitochondria and Golgi complex could not be detected (Fig. 2a, 3a,b). In addition, an increase in the number of lysosomes (Fig. 4a) was observed. The rough endoplasmic reticulum (RER) appeared disrupted. Parietal cells could be detected with difficulty due to their decrease in the number. Their cytoplasm contains many altered mitochondria and microvilli cut in all direction (Fig. 4a).
In general, electron microscopic examination of the specimens of patients infected with H. pylori visualize many structures, which cannot be seen with the light microscope. The organism itself, the irregularity of nuclei, which appear smooth by light microscope (Fig. 1), the nuclear inclusions and the nuclear pocket could be identified ultrastructurally very easily.
It was reported that, in practice detection of H. pylori is difficult in the developing world and presumptive treatment is always followed by recurrence (Guisset et al., 1997). Some investigators reported that the organisms are considered difficult to detect with convential methods and to dislodge with antimicrobial therapy (Tursi et al., 1997). However, in the present study it was noticed that, the high resolution of the electron microscope permits the Fig. 3a, identification of H. pylori easily, in the lumen of gastric gland of patients with gastrointestinal symptoms suggesting chronic gastritis.
The organisms were observed around intercellular junctions and in the parietal cell canaliculi and may affect cell function or alter gastric physiology (Taniguchi et al., 1995). These results are in agreement with our observations, since it was difficult to detect parietal cells in our preparations due to probably obvious decrease in their number. In addition, the observed parietal cells appeared morphologically altered.
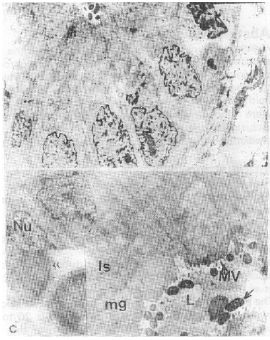 | |
| Fig. 2a, b, c: | E.M. A portion of gastric gland of patient infected with H. pylori. a). Note, the irregularity of nuclear shape and variability of heterochromatin content. Arrow pointed at shrunk pyknotic nucleus. X 5,000. b) Illustrating numerous curved-shaped organisms (arrow) in the gastric lumen (L) quite close to microvilli (Mv) of the superficial epithelial cells. Note also, paucity of cell organelles, dilated intercellular space (Is), marginating nucleolus (Nu), mucous granules (mg). X5, 000. c) Double membrane bound organism X 7,500 |
Our preparation revealed the organisms only in the gastric lumen of some glands, quite close to the microvilli. These observations are in agreement with that of Chlumska and Sedlackova (1995) who observed that the organisms most frequently appeared free in mucous layer without direct contact with the mucosa and also registered in the intracytoplasmic channels of intact parietal cells. However, in many biopsy specimens bacteria were seen closely attached to the epithelial cell membrane by different forms of adhesion. In addition to what looked like intracytoplasmic penetration by bacteria, several examples of genuine penetration were observed (Noach et al., 1994). These authors concluded that this feature might contribute to the pathogenic action of the organism.
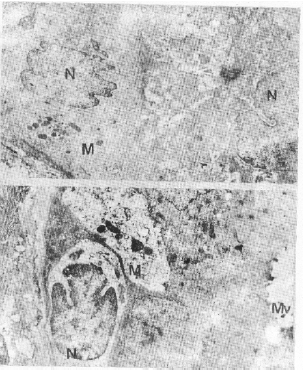 | |
| Fig. 3a, b: | E.M.Same as previous. a) Illustrating segmented nuclei (N) presents loss of chromatin. Pleomorphic aggregated mitochondria (M), altered microvilli (Mv). X, 5000.b) Demonstrating severely altered nucleus (N), aggregated mitochondria (M) vacuolated cytoplasm. X 5,000 |
Association between the organism and altered epithelial cells was noted during the present study. This is similar to the findings reported previously (Janas et al., 1995; Kobayashi et al., 1995) and showed that coccoid forms of H. pylori are closely associated with strongly damaged gastric mucous cells.
Tursi et al. (1997) reported that H. pylori, plays an essential role in the development neoplastic gastroduodenal pathologies. They stated also that, man is the only well established "reservoir" of H. pylori, while the role of other mammalians (cat, pig primates) as sources of infection is still controversy.
It is of considerable interest that our electron microscopy preparations of patients with gastritis associated with H. pylori, gastric cells revealed interesting changes at the ultrastructural level. This alteration involved both nucleus and cytoplasmic organelles, such as a high nuclear cytoplasmic ratio, pale-nuclei with a paucity of heterochromatin, deficiency of mitochondria and other cellwork-associated organelles. Ghadially (1985) reported that cells with pale nuclei and enlarged nucleoli are more important in active cell proliferation and, hence, in tumor growth. In addition, our preparations revealed nuclear irregularity in most cells of the gastric gland.
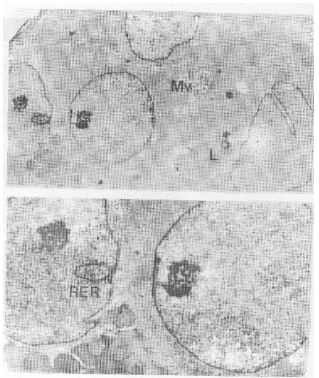 | |
| Fig. 4 a, b, c: | E.M. Same as previous, illustrating cells with large sized nuclei, the euchromatin predominate, Mv: microvilli, L: lysosome. X 5, 000. b) The arrow points to type 1 nuclear pockets, containing RER and demarcated by a band of chromatin .X 10,000. |
Nuclear irregularity is such a common phenomenon seen in tumors that is not of much value in differential diagnosis, but there are a few exceptions where the pattern of irregularity, (i.e. nuclear configuration) strongly suggests a diagnosis but does not necessarily prove it (Ghadially, 1985). Our preparation demonstrated also that gastric epithelial cells contained numerous nuclear pocket of the first type that contained cytoplasmic material. It is worthy to mentioning that an abundance of these pockets is virtually diagnostic of lymphoma or leukemia in human (Ghadially, 1985). In addition, Senoo et al. (1984) showed that type 1 nuclear pockets are produced by a fold or ruffle arising from the surface of the nucleus which entraps cytoplasmic material. However, type 2 nuclear pockets seems to arise by invagination of the inner nuclear envelope, producing a meandering cleft which sequestrates nuclear material ultimately producing a type 2 pocket. On the other hand, Abdelmeguid et al. (1997) observed the two types of nuclear pockets in amphibian tumor. Ultrastructural changes on the surface and within the cytoplasm of mucosa cells including epithelial mucous cells were observed, indicating disturbed metabolism. Similar observation was reported by Bartel et al. (1992).
It is clear from this study that the ultrastructural feature induced by H. pylori in human closely resemble those described in tumor cells.
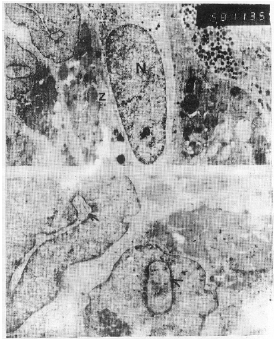 | |
| Fig. 5 a, b: | E.M. Same as previous .a) Demonstrated altered cells with large-sized nuclei (N). Z: zymogen granules. X 5,000. b) Demonstrated both nuclear pocket enclosed cytoplasmic material (arrow), nuclear inclusion containing ribosomes (arrowhead). X 7,500 |
Nevertheless, at the present stage of our knowledge about the effect of H. pylori on the epithelial cells we like to stress that the interesting point, that the frequent presence of nuclear pocket may help in early predicting the transformation of gastric cells into tumor cells.
I would like to express my deepest gratitude to Prof. Dr. Hussien A.A and Dr. Mohammed S. S, Faculty of Medicine, Alexandria University, for offering the specimens used in the study.