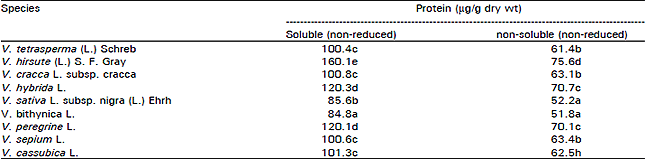
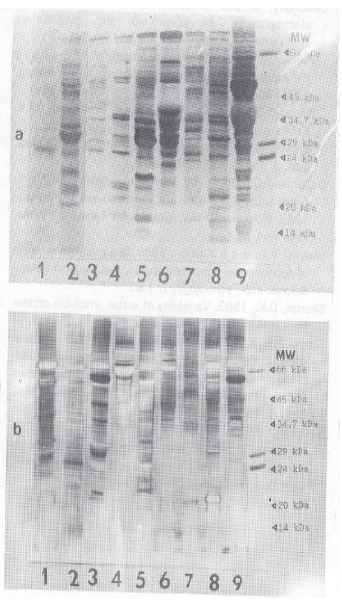
Research Article
An Electrophoretic Analysis of the Seed Proteins of some Vicia L. Species from Northeast Anatolia (Turkey)
Department of Biology, Karadeniz Technical University, 61080 Trabzon, Turkey
Huseyin Inceer
Department of Biology, Karadeniz Technical University, 61080 Trabzon, Turkey
Sema Hayirlogliu-Ayaz
Department of Biology, Karadeniz Technical University, 61080 Trabzon, Turkey