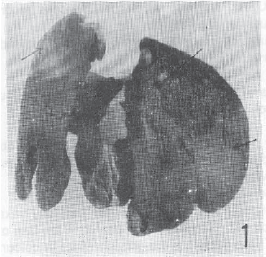
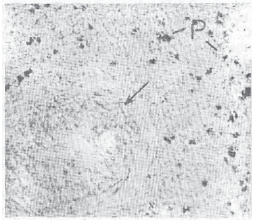
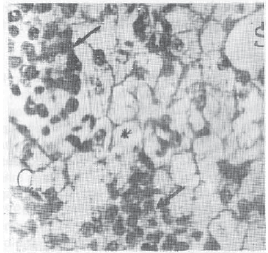
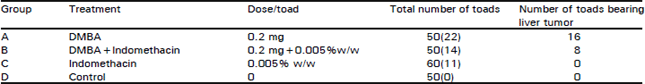
Research Article
Anti-inflammatory Drug (Indomethacin) and its Effect on Liver Tumor Induced by DMBA
Department of Zoology, Faculty of Science, Alexandria University, Alexandria, Egypt
I.A. Sadek
Department of Zoology, Faculty of Science, Alexandria University, Alexandria, Egypt
J. M. Sorour
Department of Zoology, Faculty of Science, Alexandria University, Alexandria, Egypt
A.A. Attia
Department of Zoology, Faculty of Science, Alexandria University, Alexandria, Egypt